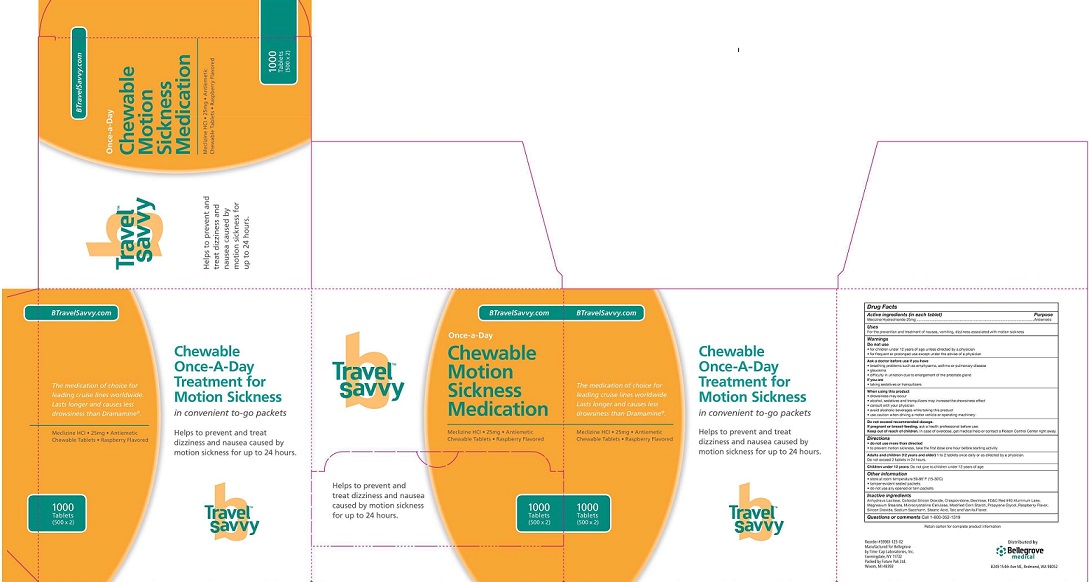
Do not use
• for children under 12 years of age unless unless directed by a physician
• for frequent or prolonged use except under the advice of a physician
Ask a doctor before use if you have
breathing problems such as emphysema, asthma or pulmonary disease
glaucoma
if you are
- taking sedatives or tranquilizers
When using this product
drowsiness may occur
alcohol, sedatives and tranquilizers may increase the drowsiness effect
consult with your physician
avoid alcoholic beverages while taking this product
use caution when driving a motor vehicle or operating machinery
Do not exceed recommended dosage
If preganant or breast-feeding, ask a health professional before use
Keep out of reach of children. In case of overdose, get medical help or contact Poison Control Center right away
Other information
- store at room temperature 59-86 degrees F (15-30 degreesC)
- tamper-evident sealed packets
- do not use any opened or torn packets
DOSAGE AND ADMINISTAATION
Directions
do not use more than directed
to provent motion sickness, take the first dose one hour before starting activity
Adults and children (12 years and older)
1 to 2 tablets once daily or as directed by a Physician
Do not exceed 2 tablets in 24 hours
Children under 12 years of age: Do not give to children under 12 years of age
INDICATION AND USAGE
For the prevention and treatment of the nausea,vomating,or dizziness associated with motion sickness.
OTC-KEEP OUT OF REACH OF CHILDREN
Do not use
• for children under 12 years of age unless unless directed by a physician
Keep out of reach of children. In case of overdose, get medical help or contact Poison Control Center right away
PURPOSE
When using this product
drowsiness may occur
alcohol, sedatives and tranquilizers may increase the drowsiness effect
consult with your physician
avoid alcoholic beverages while taking this product
use caution when driving a motor vehicle or operating machinery