Label: TEGLUTIK- riluzole liquid
- NDC Code(s): 70726-0306-1
- Packager: EDW PHARMA
- Category: HUMAN PRESCRIPTION DRUG LABEL
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated June 14, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
Important Prescribing Information
May2024
Subject: Temporary importation of TEGLUTIK (riluzole oral suspension, 5 mg/mL) with English labeling to address drug shortage in the United States
Dear Health Care Provider,
The purpose of this letter is to inform you about a temporary importation in the United States (U.S.) of TEGLUTIK (riluzole 5 mg/mL oral suspension) with bottle and carton labels in English, in coordination with the U.S. Food and Drug Administration (FDA) to mitigate the current shortage of FDA-approved Tiglutik (riluzole oral suspension, 50 mg/10 mL) in the U.S. This temporary supply of TEGLUTIK is marketed and manufactured by Italfarmaco in Spain and is not FDA-approved.
Riluzole is indicated for the treatment of amyotrophic lateral sclerosis (ALS). Recently, Tiglutik was recalled in the U.S. market due to an out-of-specification test result for viscosity.
At this time, no other entity except EDW Pharma, Inc. (formerly Italfarmaco (ITF) Pharma, Inc.) is authorized by the FDA to import or distribute Italfarmaco's TEGLUTIK riluzole oral suspension in the U.S.
Effective immediately, and during this temporary period, EDW Pharma, Inc. will distribute the following presentation of riluzole oral suspension to address the critical shortage:Product Name Quantity Descriptions U.S. NDC number Lot Number Expiration Date TEGLUTIK
riluzole oral | carton
suspension
(5 mg/mL)
1 bottle per carton Teglutik is presented
as a slightly brown,
opaque homogeneous oral
suspension after being
manually gently shaken.
TEGLUTIK is
available in a bottle
of 300 ml with a
plastic graduated oral dosing
syringe. The
syringe barrel is
graduated in
milliliters up to 10 ml.
70726-0306-1 24008 04/2027 The safety profiles of the FDA-approved Tiglutik and imported TEGLUTIK products are comparable and no specific safety concerns emerged from the comparison of the two products.
Please refer to the side-by-side comparison of the labels (enclosed) for additional information.
Tiglutik is available only by prescription in the U.S. The imported lot does not have the statement "Rx only" on its labeling.The barcode on the imported product label may not register accurately on the U.S. scanning systems. Institutions should manually input the imported product information into their systems and confirm that the barcode, if scanned, provides correct information. Alternative procedures should be followed to assure that the correct drug product is being used and administered to individual patients.
In addition, the package of the imported product does not include a product identifier as required under the Drug Supply Chain Security Act (DSCSA). Specifically, each package does not include the NDC, unique serial number, lot number, and expiration date in both human- readable and a two-dimensional data matrix barcode. Additionally, the imported product may not be accompanied with DSCSA-required product tracing documentation (transaction information, transaction history, and transaction statement).
Reporting Adverse Events
Health care providers and patients are encouraged to report adverse events and medication errors in patients taking TEGLUTIK to AnovoRx at 1-844-763-1198. You are encouraged to report negative side effects of prescription drugs to the FDA.
Adverse events or quality problems experienced with the use of this product may also be reported to the FDA's MedWatch Adverse Event Reporting Program either online, by regular mail, or by fax:
- Complete and submit the report Online: www.fda.gov/medwatch/report.htm
- Regular mail or Fax: Download form www.fda.gov/MedWatch/getforms.htm or call 1- 800-332-1088 to request a reporting form, then complete and return to the address on the pre-addressed form or submit by fax to 1-800-FDA-0178 (1-800-332-0178).
You may also contact AnovoRx at 1-844-763-1198 if you have any questions about the information contained in this letter or the safe and effective use of TEGLUTIK.
This letter is not intended as a complete description of the benefits and risks related to the use of TEGLUTIK. Please refer to the enclosed TEGLUTIK SmPC and Tiglutik USPI side-by- side comparison.
For additional information, please visit www.tiglutik.com and www.edwpharma.com.
Sincerely,
Peter Cook
CEO and President
EDW Pharma, Inc. (Formerly ITF Pharma, Inc.)
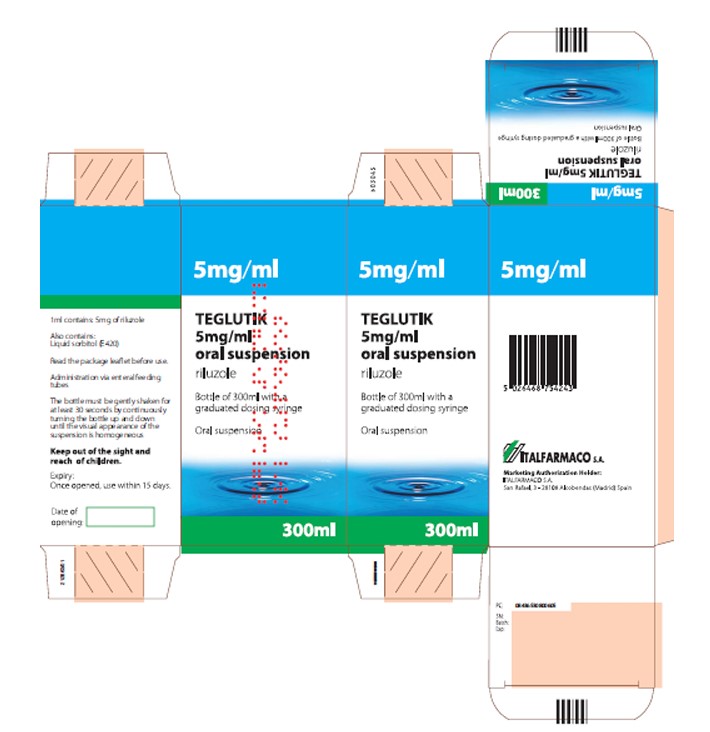

(UK Bottle TEXT)
(u.s. BOTTLE label - TEXT)
NAME OF THE MEDICINAL PRODUCT
TEGLUTIK 5 mg/ml oral suspension
riluzoleNAME OF THE MEDICINAL PRODUCT
TIGLUTIK®
riluzole
oral suspension
50mg/10ml(5mg/ml)STATEMENT OF ACTIVE SUBSTANCE(S)
1 ml contains: 5 mg of riluzole
STATEMENT OF ACTIVE SUBSTANCE(S)
Contains: TIGLUTIK® 50 mg/10 mL (5 mg/mL)
LIST OF EXCIPIENTS
Also contains: liquid sorbitol (E420)
-------
PHARMACEUTICAL FORM AND CONTENTS
Oral suspension
Bottle of 300 ml
PHARMACEUTICAL FORM AND CONTENTS
s product is a ling Information.
TIGLUTIK® 50 mg/10 mL (5 mg/mL) oral suspension is a slightly brown, opaque,
homogeneous suspension when mixed, containing 50 mg of riluzole per 10 mL of suspension
METHOD AND ROUTE(S) OF ADMINISTRATION
Read the package leaflet before use
Oral use
Administration via enteral feeding tubes
Shake gently before use
METHOD AND ROUTE(S) OF ADMINISTRATION
Before use, please read the enclosed Prescribing Information.
For oral administration
Shake gently before use
The bottle must be gently shaken for at least 30 seconds by continuously rotating the bottle 180° until the visual appearance of the suspension is homogeneous.
DOSAGE
-----
DOSAGE
The recommended dose is 10 mL of TIGLUTIK® oral suspension, containing 50 mg of riluzole, taken orally twice daily, every 12 hours.
SPECIAL WARNING THAT THE MEDICINAL PRODUCT MUST BE STORED OUT OF THE SIGHT AND REACH OF CHILDREN
Keep out of the sight and reach of children
SPECIAL WARNING THAT THE MEDICINAL PRODUCT MUST BE STORED OUT OF THE SIGHT AND REACH OF CHILDREN
Keep out of reach of children.
SPECIAL STORAGE CONDITIONS
Expiry:
Once opened, use within 15 days
SPECIAL STORAGE CONDITIONS
Store TIGLUTIK® at controlled room temperature between 20°-25°C (68°-77°F), excursions permitted to 15°-30°C (59°-86°F) and protect from bright light.
Do not freeze. Store upright.
Once opened, the bottle of TIGLUTIK® should be used within 15 days. Keep bottle tightly closed between each use.
GENERAL CLASSIFICATION FOR SUPPLY
Medicinal product subject to medical prescription
Hospital useGENERAL CLASSIFICATION FOR SUPPLY
Rx only
NAME AND ADDRESS OF THE MARKETING AUTHORISATION HOLDER
Marketing Authorization Holder:
ITALFARMACO S.A
San Rafael, 3
28108 Alcobendas (Madrid)
SpainITF Pharma
Manufactured for:
ITF Pharma, Inc.
Berwyn, PA 19312 USA
TIGLUTIK is a registered trademark of Italfarmaco S.A.
©2019 ITF Pharma, Inc. All rights reserved.
TOCXXXXXX
- PRINCIPAL DISPLAY PANEL - 300 mL Bottle Carton
-
INGREDIENTS AND APPEARANCE
TEGLUTIK
riluzole liquidProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:70726-0306 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength RILUZOLE (UNII: 7LJ087RS6F) (RILUZOLE - UNII:7LJ087RS6F) RILUZOLE 5 mg in 1 mL Inactive Ingredients Ingredient Name Strength MAGNESIUM ALUMINUM SILICATE (UNII: 6M3P64V0NC) SORBITOL (UNII: 506T60A25R) POLYOXYL 20 CETOSTEARYL ETHER (UNII: YRC528SWUY) WATER (UNII: 059QF0KO0R) SACCHARIN SODIUM (UNII: SB8ZUX40TY) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) DIMETHICONE (UNII: 92RU3N3Y1O) SODIUM LAURYL SULFATE (UNII: 368GB5141J) XANTHAN GUM (UNII: TTV12P4NEE) Product Characteristics Color brown (slightly brown, opaque) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70726-0306-1 300 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 06/10/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date Unapproved drug for use in drug shortage 06/10/2024 Labeler - EDW PHARMA (080260470) Registrant - EDW PHARMA (080260470)


