Label: ANTACID tablet
- NDC Code(s): 83698-002-01
- Packager: Xiamen Kang Zhongyuan Biotechnology Co., Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated May 4, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
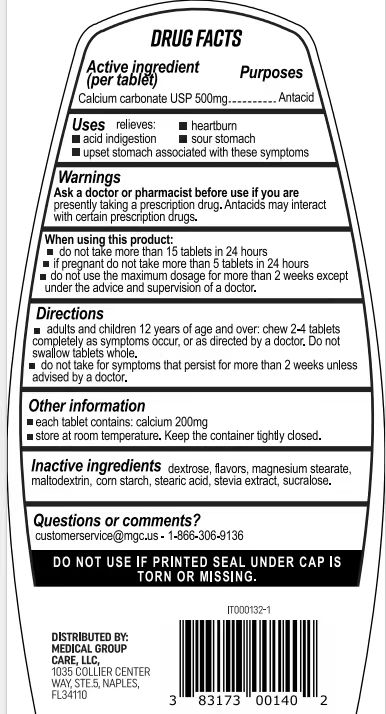
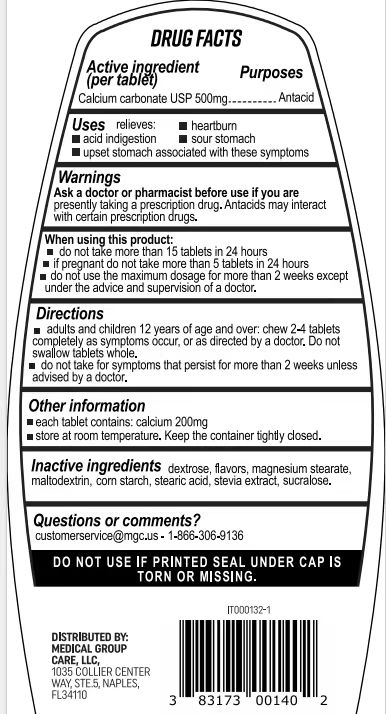
- Active Ingredient
- Purpose
- Use
- Warnings
- Do not use
- When Using
- Keep Oot Of Reach Of Children
- Directions
- Other information
- Inactive ingredients
- Questions
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ANTACID
antacid tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:83698-002 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CALCIUM CARBONATE (UNII: H0G9379FGK) (CALCIUM CATION - UNII:2M83C4R6ZB) CALCIUM CARBONATE 500 mg in 1 g Inactive Ingredients Ingredient Name Strength ICODEXTRIN (UNII: 2NX48Z0A9G) MAGNESIUM STEARATE (UNII: 70097M6I30) ANHYDROUS DEXTROSE (UNII: 5SL0G7R0OK) Product Characteristics Color white Score no score Shape ROUND Size 3mm Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:83698-002-01 100 g in 1 BOTTLE; Type 0: Not a Combination Product 05/05/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M001 05/05/2024 Labeler - Xiamen Kang Zhongyuan Biotechnology Co., Ltd. (411759931) Establishment Name Address ID/FEI Business Operations Xiamen Kang Zhongyuan Biotechnology Co., Ltd. 411759931 manufacture(83698-002)