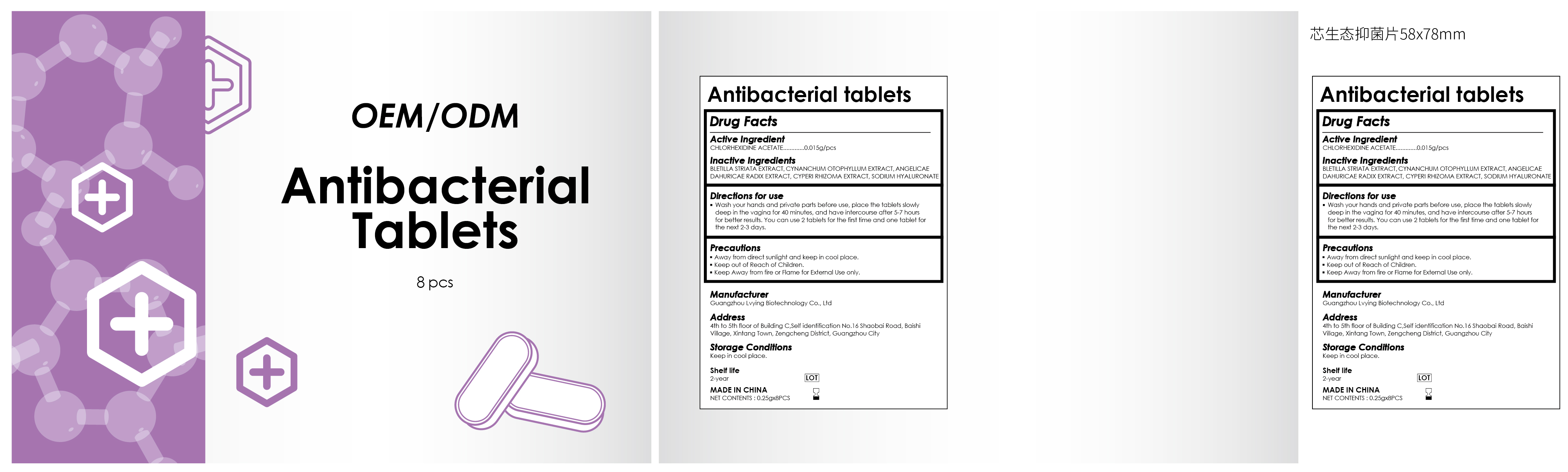
Label: ANTIBACTERIALTABLETS- chlorhexidine acetate tablet
- NDC Code(s): 84260-005-01
- Packager: Guangzhou Lvying Biotechnology Co., Ltd
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated May 16, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
- WARNINGS
- DOSAGE & ADMINISTRATION
- STORAGE AND HANDLING
- INACTIVE INGREDIENT
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ANTIBACTERIALTABLETS
chlorhexidine acetate tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:84260-005 Route of Administration VAGINAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CHLORHEXIDINE ACETATE (UNII: 5908ZUF22Y) (CHLORHEXIDINE - UNII:R4KO0DY52L) CHLORHEXIDINE ACETATE 0.015 g Inactive Ingredients Ingredient Name Strength HYALURONATE SODIUM (UNII: YSE9PPT4TH) ANGELICA DAHURICA ROOT (UNII: 1V63N2S972) CYPERUS ROTUNDUS TUBER (UNII: 4B51SRR959) BLETILLA STRIATA WHOLE (UNII: WV2BSG60EW) CYNANCHUM OTOPHYLLUM WHOLE (UNII: 02I89C6K47) Product Characteristics Color white Score score with uneven pieces Shape OVAL Size 13mm Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:84260-005-01 8 in 1 BOX; Type 0: Not a Combination Product 04/29/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug 505G(a)(3) 04/29/2024 Labeler - Guangzhou Lvying Biotechnology Co., Ltd (714445262) Establishment Name Address ID/FEI Business Operations Guangzhou Lvying Biotechnology Co., Ltd 714445262 manufacture(84260-005)