Label: LANTUS- insulin glargine injection, solution
LANTUS SOLOSTAR- insulin glargine injection, solution
-
NDC Code(s):
0088-2219-00,
0088-2219-01,
0088-2219-05,
0088-2220-33, view more0088-2220-34
- Packager: sanofi-aventis U.S. LLC
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Biologic Licensing Application
Drug Label Information
Updated May 23, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use LANTUS safely and effectively. See full prescribing information for LANTUS.
LANTUS® (insulin glargine) injection, for subcutaneous use
Initial U.S. Approval: 2000INDICATIONS AND USAGE
DOSAGE AND ADMINISTRATION
- Individualize dosage based on metabolic needs, blood glucose monitoring, glycemic control, type of diabetes, and prior insulin use. (2.2)
- Administer subcutaneously into the abdominal area, thigh, or deltoid once daily at any time of day, but at the same time every day. (2.1)
- Do not dilute or mix with any other insulin or solution. (2.1)
- Rotate injection sites to reduce risk of lipodystrophy and localized cutaneous amyloidosis. (2.1)
- See Full Prescribing Information for the recommended starting dosage in patients with type 2 diabetes (2.3) and how to change to LANTUS from other insulins (2.4)
- Closely monitor glucose when switching to LANTUS and during initial weeks thereafter. (2.4)
DOSAGE FORMS AND STRENGTHS
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
- Never share a LANTUS SoloStar prefilled pen, insulin syringe, or needle between patients, even if the needle is changed. (5.1)
- Hyperglycemia or hypoglycemia with changes in insulin regimen: Make changes to a patient's insulin regimen (e.g., insulin strength, manufacturer, type, injection site or method of administration) under close medical supervision with increased frequency of blood glucose monitoring. (5.2)
- Hypoglycemia: May be life-threatening. Increase frequency of glucose monitoring with changes to: insulin dosage, concomitant drugs, meal pattern, physical activity; and in patients with renal or hepatic impairment and hypoglycemia unawareness. (5.3)
- Hypoglycemia due to medication errors: Accidental mix-ups between insulin products can occur. Instruct patients to check insulin labels before injection. (5.4)
- Hypersensitivity reactions: Severe, life-threatening, generalized allergy, including anaphylaxis, can occur. Discontinue LANTUS. Monitor and treat if indicated. (5.5)
- Hypokalemia: May be life-threatening. Monitor potassium levels in patients at risk of hypokalemia and treat if indicated. (5.6)
- Fluid retention and heart failure with concomitant use of thiazolidinediones (TZDs): Observe for signs and symptoms of heart failure; consider dosage reduction or discontinuation of TZD if heart failure occurs. (5.7)
ADVERSE REACTIONS
Adverse reactions commonly associated with LANTUS include hypoglycemia, allergic reactions, injection site reactions, lipodystrophy, pruritus, rash, edema, and weight gain. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact sanofi-aventis at 1-800-633-1610 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.DRUG INTERACTIONS
See 17 for PATIENT COUNSELING INFORMATION, FDA-approved patient labeling and FDA-approved patient labeling.
Revised: 6/2023
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Important Administration Instructions
2.2 General Dosing Instructions
2.3 Initiation of LANTUS Therapy
2.4 Switching to LANTUS from Other Insulin Therapies
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Never Share a LANTUS SoloStar Prefilled Pen, Insulin Syringe, or Needle Between Patients
5.2 Hyperglycemia or Hypoglycemia with Changes in Insulin Regimen
5.3 Hypoglycemia
5.4 Hypoglycemia Due to Medication Errors
5.5 Hypersensitivity Reactions
5.6 Hypokalemia
5.7 Fluid Retention and Heart Failure with Concomitant Use of PPAR-gamma Agonists
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Immunogenicity
6.3 Postmarketing Experience
7 DRUG INTERACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
8.7 Hepatic Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
14.1 Overview of Clinical Studies
14.2 Clinical Studies in Adult and Pediatric Patients with Type 1 Diabetes
14.3 Clinical Studies in Adults with Type 2 Diabetes
14.4 Additional Clinical Studies in Adults with Diabetes Type 1 and Type 2
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
16.2 Storage
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
2.1 Important Administration Instructions
- Always check insulin labels before administration [see Warnings and Precautions (5.4)]
- Visually inspect LANTUS vials and SoloStar prefilled pens for particulate matter and discoloration prior to administration. Only use if the solution is clear and colorless with no visible particles.
- Administer LANTUS subcutaneously into the abdominal area, thigh, or deltoid, and rotate injection sites within the same region from one injection to the next to reduce the risk of lipodystrophy and localized cutaneous amyloidosis. Do not inject into areas of lipodystrophy or localized cutaneous amyloidosis [see Warnings and Precautions (5.2) and Adverse Reactions (6)].
- During changes to a patient's insulin regimen, increase the frequency of blood glucose monitoring [see Warnings and Precautions (5.2)].
- Do not administer intravenously or via an insulin pump.
- Do not dilute or mix LANTUS with any other insulin or solution.
- The LANTUS SoloStar prefilled pen dials in 1-unit increments.
Use LANTUS SoloStar prefilled pen with caution in patients with visual impairment who may rely on audible clicks to dial their dose.
2.2 General Dosing Instructions
- Administer LANTUS subcutaneously once daily at any time of day but at the same time every day.
- Individualize and adjust the dosage of LANTUS based on the patient's metabolic needs, blood glucose monitoring results and glycemic control goal.
- Dosage adjustments may be needed with changes in physical activity, changes in meal patterns (i.e., macronutrient content or timing of food intake), during acute illness, or changes in renal or hepatic function. Dosage adjustments should only be made under medical supervision with appropriate glucose monitoring [see Warnings and Precautions (5.2)].
- In patients with type 1 diabetes, LANTUS must be used concomitantly with short-acting insulin.
2.3 Initiation of LANTUS Therapy
2.4 Switching to LANTUS from Other Insulin Therapies
Dosage adjustments are recommended to lower the risk of hypoglycemia when switching patients to LANTUS from other insulin therapies [see Warnings and Precautions (5.3)].
When switching from:
- Once-daily TOUJEO (insulin glargine 300 units/mL) to once-daily LANTUS (100 units/mL), the recommended starting LANTUS dosage is 80% of the TOUJEO dosage that is being discontinued.
- Once-daily NPH insulin to once-daily LANTUS, the recommended starting LANTUS dosage is the same as the dosage of NPH that is being discontinued.
- Twice-daily NPH insulin to once-daily LANTUS, the recommended starting LANTUS dosage is 80% of the total NPH dosage that is being discontinued.
- 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
LANTUS is contraindicated:
- During episodes of hypoglycemia [see Warnings and Precautions (5.3)]
- In patients with hypersensitivity to insulin glargine or any of the excipients in LANTUS [see Warnings and Precautions (5.5)]
-
5 WARNINGS AND PRECAUTIONS
5.1 Never Share a LANTUS SoloStar Prefilled Pen, Insulin Syringe, or Needle Between Patients
LANTUS SoloStar prefilled pens must never be shared between patients, even if the needle is changed. Patients using LANTUS vials must never re-use or share needles or syringes with another person. Sharing poses a risk for transmission of blood-borne pathogens.
5.2 Hyperglycemia or Hypoglycemia with Changes in Insulin Regimen
Changes in an insulin regimen (e.g., insulin strength, manufacturer, type, injection site or method of administration) may affect glycemic control and predispose to hypoglycemia [see Warnings and Precautions (5.3)] or hyperglycemia. Repeated insulin injections into areas of lipodystrophy or localized cutaneous amyloidosis have been reported to result in hyperglycemia; and a sudden change in the injection site (to unaffected area) has been reported to result in hypoglycemia [see Adverse Reactions (6)].
Make any changes to a patient's insulin regimen under close medical supervision with increased frequency of blood glucose monitoring. Advise patients who have repeatedly injected into areas of lipodystrophy or localized cutaneous amyloidosis to change the injection site to unaffected areas and closely monitor for hypoglycemia. For patients with type 2 diabetes, dosage adjustments of concomitant oral and antidiabetic products may be needed.
5.3 Hypoglycemia
Hypoglycemia is the most common adverse reaction associated with insulins, including LANTUS. Severe hypoglycemia can cause seizures, may be life-threatening or cause death. Hypoglycemia can impair concentration ability and reaction time; this may place the patient and others at risk in situations where these abilities are important (e.g., driving or operating other machinery).
Hypoglycemia can happen suddenly, and symptoms may differ in each patient and change over time in the same patient. Symptomatic awareness of hypoglycemia may be less pronounced in patients with longstanding diabetes, in patients with diabetic neuropathy, using drugs that block the sympathetic nervous system (e.g., beta-blockers) [see Drug Interactions (7)], or who experience recurrent hypoglycemia.
The long-acting effect of LANTUS may delay recovery from hypoglycemia.
Risk Factors for Hypoglycemia
The risk of hypoglycemia after an injection is related to the duration of action of the insulin and, in general, is highest when the glucose lowering effect of the insulin is maximal. As with all insulins, the glucose lowering effect time course of LANTUS may vary in different patients or at different times in the same patient and depends on many conditions, including the area of injection as well as the injection site blood supply and temperature [see Clinical Pharmacology (12.2)]. Other factors which may increase the risk of hypoglycemia include changes in meal pattern (e.g., macronutrient content or timing of meals), changes in level of physical activity, or changes to concomitant drugs [see Drug Interactions (7)]. Patients with renal or hepatic impairment may be at higher risk of hypoglycemia [see Use in Specific Populations (8.6, 8.7)].
Risk Mitigation Strategies for Hypoglycemia
Patients and caregivers must be educated to recognize and manage hypoglycemia. Self-monitoring of blood glucose plays an essential role in the prevention and management of hypoglycemia. In patients at higher risk for hypoglycemia and patients who have reduced symptomatic awareness of hypoglycemia, increased frequency of blood glucose monitoring is recommended.
5.4 Hypoglycemia Due to Medication Errors
Accidental mix-ups among insulin products have been reported. To avoid medication errors between LANTUS and other insulins, instruct patients to always check the insulin label before each injection [see Adverse Reactions (6.3)].
5.5 Hypersensitivity Reactions
Severe, life-threatening, generalized allergy, including anaphylaxis, can occur with insulins, including LANTUS [see Adverse Reactions (6.1)]. If hypersensitivity reactions occur, discontinue LANTUS; treat per standard of care and monitor until symptoms and signs resolve. LANTUS is contraindicated in patients who have had hypersensitivity reactions to insulin glargine or one of the excipients.
5.6 Hypokalemia
All insulins, including LANTUS, cause a shift in potassium from the extracellular to intracellular space, possibly leading to hypokalemia. Untreated hypokalemia may cause respiratory paralysis, ventricular arrhythmia, and death. Monitor potassium levels in patients at risk for hypokalemia, if indicated (e.g., patients using potassium-lowering medications, patients taking medications sensitive to serum potassium concentrations).
5.7 Fluid Retention and Heart Failure with Concomitant Use of PPAR-gamma Agonists
Thiazolidinediones (TZDs), which are peroxisome proliferator-activated receptor (PPAR)-gamma agonists, can cause dose-related fluid retention, when used in combination with insulin. Fluid retention may lead to or exacerbate heart failure. Patients treated with insulin, including LANTUS, and a PPAR-gamma agonist should be observed for signs and symptoms of heart failure. If heart failure develops, it should be managed according to current standards of care, and discontinuation or dose reduction of the PPAR-gamma agonist must be considered.
-
6 ADVERSE REACTIONS
The following adverse reactions are discussed elsewhere:
- Hyperglycemia or Hypoglycemia with Changes in Insulin Regimen [see Warnings and Precautions (5.2)]
- Hypoglycemia [see Warnings and Precautions (5.3)]
- Hypoglycemia Due to Medication Errors [see Warnings and Precautions (5.4)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.5)]
- Hypokalemia [see Warnings and Precautions (5.6)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in clinical trials of a drug cannot be directly compared to rates in the clinical trial of another drug and may not reflect the rates observed in practice.
The data in Table 1 reflect the exposure of 2,327 patients with type 1 diabetes to LANTUS or NPH in Studies A, B, C, and D [see Clinical Studies (14.2)]. The type 1 diabetes population had the following characteristics: the mean age was 39 years, 54% were male, and mean body mass index (BMI) was 25.1 kg/m2. Ninety-seven percent were White, 2% were Black or African American and less than 1% were Asian. Approximately 3% of the patients in studies B and C were Hispanic.
The data in Table 2 reflect the exposure of 1,563 patients with type 2 diabetes to LANTUS or NPH in Studies E, F, and G [see Clinical Studies (14.3)]. The type 2 diabetes population had the following characteristics: the mean age was 59 years, 58% were male, and mean BMI was 29.2 kg/m2. Eighty-seven percent were White, 8% were Black or African American and 3% were Asian. Approximately 9% of patients in Study F were Hispanic.
The frequencies of adverse reactions during LANTUS clinical studies in patients with type 1 diabetes mellitus and type 2 diabetes mellitus are listed in the tables below (Tables 1, 2, 3, and 4).
Table 1: Adverse Reactions Occurring ≥5% in Pooled Clinical Studies up to 28 Weeks Duration in Adults with Type 1 Diabetes LANTUS, %
(n=1,257)NPH, %
(n=1,070)- *
- Body system not specified
Upper respiratory tract infection 22.4 23.1 Infection* 9.4 10.3 Accidental injury 5.7 6.4 Headache 5.5 4.7 Table 2: Adverse Reactions Occurring ≥5% in Pooled Clinical Studies up to 1 Year Duration in Adults with Type 2 Diabetes LANTUS, %
(n=849)NPH, %
(n=714)- *
- Body system not specified
Upper respiratory tract infection 11.4 13.3 Infection* 10.4 11.6 Retinal vascular disorder 5.8 7.4 Table 3: Adverse Reactions Occurring ≥10% in a 5-Year Study of Adults with Type 2 Diabetes LANTUS, %
(n=514)NPH, %
(n=503)Upper respiratory tract infection 29.0 33.6 Edema peripheral 20.0 22.7 Hypertension 19.6 18.9 Influenza 18.7 19.5 Sinusitis 18.5 17.9 Cataract 18.1 15.9 Bronchitis 15.2 14.1 Arthralgia 14.2 16.1 Pain in extremity 13.0 13.1 Back pain 12.8 12.3 Cough 12.1 7.4 Urinary tract infection 10.7 10.1 Diarrhea 10.7 10.3 Depression 10.5 9.7 Headache 10.3 9.3 Table 4: Adverse Reactions Occurring ≥5% in a 28-Week Clinical Study in Pediatric Patients with Type 1 Diabetes LANTUS, %
(n=174)NPH, %
(n=175)- *
- Body system not specified
Infection* 13.8 17.7 Upper respiratory tract infection 13.8 16.0 Pharyngitis 7.5 8.6 Rhinitis 5.2 5.1 Severe Hypoglycemia
Hypoglycemia was the most commonly observed adverse reaction in patients treated with LANTUS. Tables 5, 6, and 7 summarize the incidence of severe hypoglycemia in the LANTUS clinical studies. Severe symptomatic hypoglycemia was defined as an event with symptoms consistent with hypoglycemia requiring the assistance of another person and associated with either a blood glucose below 50 mg/dL (≤56 mg/dL in the 5-year study and ≤36 mg/dL in the ORIGIN study) or prompt recovery after oral carbohydrate, intravenous glucose, or glucagon administration.
Percentages of LANTUS-treated adult patients who experienced severe symptomatic hypoglycemia in the LANTUS clinical studies [see Clinical Studies (14)] were comparable to percentages of NPH-treated patients for all treatment regimens (see Tables 5 and 6). In the pediatric clinical study, pediatric patients with type 1 diabetes had a higher incidence of severe symptomatic hypoglycemia in the two treatment groups compared to the adult studies with type 1 diabetes.
Table 5: Severe Symptomatic Hypoglycemia in Patients with Type 1 Diabetes Study A
Type 1 Diabetes
Adults
28 weeks
In combination with regular insulinStudy B
Type 1 Diabetes
Adults
28 weeks
In combination with regular insulinStudy C
Type 1 Diabetes
Adults
16 weeks
In combination with insulin lisproStudy D
Type 1 Diabetes
Pediatrics
26 weeks
In combination with regular insulinLANTUS
N=292NPH
N=293LANTUS
N=264NPH
N=270LANTUS
N=310NPH
N=309LANTUS
N=174NPH
N=175Percent of patients 10.6 15.0 8.7 10.4 6.5 5.2 23.0 28.6 Table 6: Severe Symptomatic Hypoglycemia in Patients with Type 2 Diabetes Study E
Type 2 Diabetes
Adults
52 weeks
In combination with oral agentsStudy F
Type 2 Diabetes
Adults
28 weeks
In combination with regular insulinStudy G
Type 2 Diabetes
Adults
5 years
In combination with regular insulinLANTUS
N=289NPH
N=281LANTUS
N=259NPH
N=259LANTUS
N=513NPH
N=504Percent of patients 1.7 1.1 0.4 2.3 7.8 11.9 Table 7 displays the proportion of patients who experienced severe symptomatic hypoglycemia in the LANTUS and Standard Care groups in the ORIGIN study [see Clinical Studies (14)].
Table 7: Severe Symptomatic Hypoglycemia in the ORIGIN Study ORIGIN Study
Median duration of follow-up: 6.2 yearsLANTUS
N=6231Standard Care
N=6273Percent of patients 5.6 1.8 Peripheral Edema
Some patients taking LANTUS have experienced sodium retention and edema, particularly if previously poor metabolic control was improved by intensified insulin therapy.
Lipodystrophy
Administration of insulin subcutaneously, including LANTUS, has resulted in lipoatrophy (depression in the skin) or lipohypertrophy (enlargement or thickening of tissue) in some patients [see Dosage and Administration (2.2)].
Insulin Initiation and Intensification of Glucose Control
Intensification or rapid improvement in glucose control has been associated with a transitory, reversible ophthalmologic refraction disorder, worsening of diabetic retinopathy, and acute painful peripheral neuropathy. However, long-term glycemic control decreases the risk of diabetic retinopathy and neuropathy.
Weight Gain
Weight gain has occurred with insulin including LANTUS and has been attributed to the anabolic effects of insulin and the decrease in glucosuria.
Hypersensitivity Reactions
Local Reactions
Patients taking LANTUS experienced injection site reactions, including redness, pain, itching, urticaria, edema, and inflammation. In clinical studies in adult patients, there was a higher incidence of injection site pain in LANTUS-treated patients (2.7%) compared to NPH insulin-treated patients (0.7%). The reports of pain at the injection site did not result in discontinuation of therapy.
6.2 Immunogenicity
As with all therapeutic proteins, there is potential for immunogenicity. All insulin products can elicit the formation of insulin antibodies. The presence of such insulin antibodies may increase or decrease the efficacy of insulin and may require adjustment of the insulin dose. In clinical studies of LANTUS, increases in titers of antibodies to insulin were observed in NPH insulin and LANTUS treatment groups with similar incidences.
6.3 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of LANTUS. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Medication errors have been reported in which rapid-acting insulins and other insulins, have been accidentally administered instead of LANTUS.
Localized cutaneous amyloidosis at the injection site has occurred. Hyperglycemia has been reported with repeated insulin injections into areas of localized cutaneous amyloidosis; hypoglycemia has been reported with a sudden change to an unaffected injection site.
-
7 DRUG INTERACTIONS
Table 8 includes clinically significant drug interactions with LANTUS.
Table 8: Clinically Significant Drug Interactions with LANTUS Drugs that May Increase the Risk of Hypoglycemia Drugs: Antidiabetic agents, ACE inhibitors, angiotensin II receptor blocking agents, disopyramide, fibrates, fluoxetine, monoamine oxidase inhibitors, pentoxifylline, pramlintide, salicylates, somatostatin analogs (e.g., octreotide), sulfonamide antibiotics. GLP-1 receptor agonists, DPP-4 inhibitors, and SGLT-2 inhibitors. Intervention: Dosage reductions and increased frequency of glucose monitoring may be required when LANTUS is coadministered with these drugs. Drugs that May Decrease the Blood Glucose Lowering Effect of LANTUS Drugs: Atypical antipsychotics (e.g., olanzapine and clozapine), corticosteroids, danazol, diuretics, estrogens, glucagon, isoniazid, niacin, oral contraceptives, phenothiazines, progestogens (e.g., in oral contraceptives), protease inhibitors, somatropin, sympathomimetic agents (e.g., albuterol, epinephrine, terbutaline), and thyroid hormones. Intervention: Dosage increases and increased frequency of glucose monitoring may be required when LANTUS is coadministered with these drugs. Drugs that May Increase or Decrease the Blood Glucose Lowering Effect of LANTUS Drugs: Alcohol, beta-blockers, clonidine, and lithium salts. Pentamidine may cause hypoglycemia, which may sometimes be followed by hyperglycemia. Intervention: Dosage adjustment and increased frequency of glucose monitoring may be required when LANTUS is coadministered with these drugs. Drugs that May Blunt Signs and Symptoms of Hypoglycemia Drugs: Beta-blockers, clonidine, guanethidine, and reserpine. Intervention: Increased frequency of glucose monitoring may be required when LANTUS is coadministered with these drugs. -
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Published studies with use of insulin glargine during pregnancy have not reported a clear association with insulin glargine and adverse developmental outcomes (see Data). There are risks to the mother and fetus associated with poorly controlled diabetes in pregnancy (see Clinical Considerations).
Rats and rabbits were exposed to insulin glargine in animal reproduction studies during organogenesis, respectively 50 times and 10 times the human subcutaneous dosage of 0.2 units/kg/day. Overall, the effects of insulin glargine did not generally differ from those observed with regular human insulin (see Data).
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively. The estimated background risk of major birth defects is 6% to 10% in women with pregestational diabetes with a peri-conceptional HbA1c >7 and has been reported to be as high as 20% to 25% in women with a peri-conceptional HbA1c >10. The estimated background risk of miscarriage for the indicated population is unknown.
Clinical Considerations
Disease-Associated Maternal and/or Embryo-fetal Risk
Hypoglycemia and hyperglycemia occur more frequently during pregnancy in patients with pre-gestational diabetes. Poorly controlled diabetes in pregnancy increases the maternal risk for diabetic ketoacidosis, preeclampsia, spontaneous abortions, preterm delivery, and delivery complications. Poorly controlled diabetes increases the fetal risk for major birth defects, stillbirth, and macrosomia-related morbidity.
Data
Human Data
Published data do not report a clear association with insulin glargine and major birth defects, miscarriage, or adverse maternal or fetal outcomes when insulin glargine is used during pregnancy. However, these studies cannot definitely establish the absence of any risk because of methodological limitations including small sample size and some lacking comparator groups.
Animal Data
Subcutaneous reproduction and teratology studies have been performed with insulin glargine and regular human insulin in rats and Himalayan rabbits. Insulin glargine was given to female rats before mating, during mating, and throughout pregnancy at doses up to 0.36 mg/kg/day, which is approximately 50 times the recommended human subcutaneous starting dosage of 0.2 units/kg/day (0.007 mg/kg/day), on a mg/kg basis. In rabbits, doses of 0.072 mg/kg/day, which is approximately 10 times the recommended human subcutaneous starting dosage of 0.2 units/kg/day on a mg/kg basis, were administered during organogenesis. The effects of insulin glargine did not generally differ from those observed with regular human insulin in rats or rabbits. However, in rabbits, five fetuses from two litters of the high-dose group exhibited dilation of the cerebral ventricles. Fertility and early embryonic development appeared normal.
8.2 Lactation
Risk Summary
There are either no or only limited data on the presence of insulin glargine in human milk, the effects on breastfed infant, or the effects on milk production. Endogenous insulin is present in human milk. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for LANTUS, and any potential adverse effects on the breastfed child from LANTUS or from the underlying maternal condition.
8.4 Pediatric Use
The safety and effectiveness of LANTUS to improve glycemic control in pediatric patients with diabetes mellitus have been established. Use of LANTUS for this indication is supported by evidence from an adequate and well-controlled study (Study D) in 174 LANTUS-treated pediatric patients aged 6 to 15 years with type 1 diabetes mellitus, and from adequate and well-controlled studies of LANTUS in adults with diabetes mellitus [see Clinical Pharmacology (12.3), Clinical Studies (14.2)].
In the pediatric clinical study, pediatric patients with type 1 diabetes had a higher incidence of severe symptomatic hypoglycemia compared to the adults in studies with type 1 diabetes [see Adverse Reactions (6.1)].
8.5 Geriatric Use
Of the total number of subjects in controlled clinical studies of patients with type 1 and type 2 diabetes who were treated with LANTUS, 15% (n=316) were ≥65 years of age and 2% (n=42) were ≥75 years of age. No overall differences in safety or effectiveness of LANTUS have been observed between patients 65 years of age and older and younger adult patients.
Nevertheless, caution should be exercised when LANTUS is administered to geriatric patients. In geriatric patients with diabetes, the initial dosing, dosage increments, and maintenance dosage should be conservative to avoid hypoglycemic reactions. Hypoglycemia may be difficult to recognize in geriatric patients.
8.6 Renal Impairment
The effect of kidney impairment on the pharmacokinetics of LANTUS has not been studied. Some studies with human insulin have shown increased circulating levels of insulin in patients with kidney failure. Frequent glucose monitoring and dosage adjustment may be necessary for LANTUS in patients with kidney impairment [see Warnings and Precautions (5.3)].
8.7 Hepatic Impairment
The effect of hepatic impairment on the pharmacokinetics of LANTUS has not been studied. Frequent glucose monitoring and dosage adjustment may be necessary for LANTUS in patients with hepatic impairment [see Warnings and Precautions (5.3)].
-
10 OVERDOSAGE
Excess insulin administration may cause hypoglycemia and hypokalemia [see Warnings and Precautions (5.3, 5.6)].
Mild episodes of hypoglycemia can usually be treated with oral carbohydrates. Lowering the insulin dosage, and adjustments in meal patterns or exercise may be needed.
More severe episodes of hypoglycemia with coma, seizure, or neurologic impairment may be treated with glucagon for emergency use or concentrated intravenous glucose. After apparent clinical recovery from hypoglycemia, continued observation and additional carbohydrate intake may be necessary to avoid recurrence of hypoglycemia.
Hypokalemia must be corrected appropriately.
-
11 DESCRIPTION
Insulin glargine is a long-acting human insulin analog produced by recombinant DNA technology utilizing a non-pathogenic laboratory strain of Escherichia coli (K12). Insulin glargine differs from human insulin in that the amino acid asparagine at position A21 is replaced by glycine and two arginines are added to the C-terminus of the B-chain. Insulin glargine has a molecular weight of 6063 Da.
LANTUS (insulin glargine) injection is a sterile, clear and colorless solution for subcutaneous use in a 10 mL multiple-dose vial or a 3 mL single-patient use prefilled pen (LANTUS Solostar).
Prefilled Pen (LANTUS Solostar) and Vial: Each mL contains 100 units of insulin glargine and the inactive ingredients: glycerol 85% (20 mg), m-cresol (2.7 mg), zinc (30 mcg), and Water for Injection, USP. The vial also contains polysorbate 20 (20 mcg). The pH is adjusted by addition of aqueous solutions of hydrochloric acid and sodium hydroxide. LANTUS has a pH of approximately 4.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
The primary activity of insulin, including insulin glargine, is regulation of glucose metabolism. Insulin and its analogs lower blood glucose by stimulating peripheral glucose uptake, especially by skeletal muscle and fat, and by inhibiting hepatic glucose production. Insulin inhibits lipolysis and proteolysis, and enhances protein synthesis.
12.2 Pharmacodynamics
In clinical studies, the glucose-lowering effect on a molar basis (i.e., when given at the same doses) of intravenous LANTUS is approximately the same as that for human insulin. Figure 1 shows results from a study in patients with type 1 diabetes conducted for a maximum of 24 hours after subcutaneous injection of LANTUS or NPH insulin. The median time between subcutaneous injection and the end of pharmacological effect was 14.5 hours (range: 9.5 to 19.3 hours) for NPH insulin, and 24 hours (range: 10.8 to >24 hours) (24 hours was the end of the observation period) for LANTUS.
* Determined as amount of glucose infused to maintain constant plasma glucose levels Figure 1: Glucose-Lowering Effect Over 24 Hours in Patients with Type 1 Diabetes 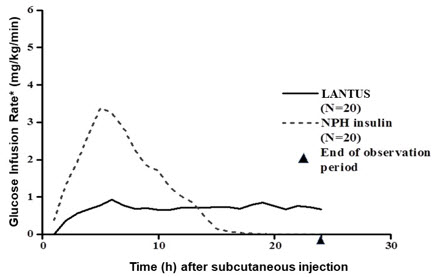
The duration of action after abdominal, deltoid, or thigh subcutaneous administration of LANTUS was similar. The time course of action of insulins, including LANTUS, may vary between patients and within the same patient.
12.3 Pharmacokinetics
Absorption
After subcutaneous injection of LANTUS in healthy subjects and in patients with diabetes, the insulin serum concentrations indicated a slower, more prolonged absorption and a relatively constant concentration/time profile over 24 hours with no pronounced peak in comparison to NPH insulin.
Elimination
Metabolism
A metabolism study in humans indicates that insulin glargine is partly metabolized at the carboxyl terminus of the B chain in the subcutaneous depot to form two active metabolites with in vitro activity similar to that of human insulin, M1 (21A-Gly-insulin) and M2 (21A-Gly-des-30B-Thr-insulin). Unchanged drug and these degradation products are also present in the circulation.
Specific Populations
Age, Race, Body Mass Index, and Gender
Effect of age, race, body mass index (BMI), and gender on the pharmacokinetics of insulin glargine has not been evaluated. However, in controlled clinical studies in adults (n=3,890) and a controlled clinical study in pediatric patients (n=349), subgroup analyses based on age, race, BMI, and gender did not show differences in safety and efficacy between LANTUS and NPH insulin [see Clinical Studies (14)].
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In mice and rats, standard two-year carcinogenicity studies with insulin glargine were performed at doses up to 0.455 mg/kg, which was for the rat approximately 65 times the recommended human subcutaneous starting dosage of 0.2 units/kg/day (0.007 mg/kg/day) on a mg/kg basis. Histiocytomas were found at injection sites in male rats and mice in acid vehicle containing groups and are considered a response to chronic tissue irritation and inflammation in rodents. These tumors were not found in female animals, in saline control, or insulin comparator groups using a different vehicle.
Insulin glargine was not mutagenic in tests for detection of gene mutations in bacteria and mammalian cells (Ames and HGPRT-test) and in tests for detection of chromosomal aberrations (cytogenetics in vitro in V79 cells and in vivo in Chinese hamsters).
In a combined fertility and prenatal and postnatal study in male and female rats at subcutaneous doses up to 0.36 mg/kg/day, which was approximately 50 times the recommended human subcutaneous starting dose of 0.2 units/kg/day (0.007 mg/kg/day) maternal toxicity due to dose-dependent hypoglycemia, including some deaths, was observed. Consequently, a reduction of the rearing rate occurred in the high-dose group only. Similar effects were observed with NPH insulin.
-
14 CLINICAL STUDIES
14.1 Overview of Clinical Studies
The safety and effectiveness of LANTUS given once-daily at bedtime was compared to that of once-daily and twice-daily NPH insulin in open-label, randomized, active-controlled, parallel studies of 2,327 adult patients and 349 pediatric patients with type 1 diabetes mellitus and 1,563 adult patients with type 2 diabetes mellitus (see Tables 9–11). In general, the reduction in glycated hemoglobin (HbA1c) with LANTUS was similar to that with NPH insulin.
14.2 Clinical Studies in Adult and Pediatric Patients with Type 1 Diabetes
Adult Patients with Type 1 Diabetes
In two clinical studies (Studies A and B), adult patients with type 1 diabetes (Study A, n=585, Study B n=534) were randomized to 28 weeks of basal-bolus treatment with LANTUS or NPH insulin. Regular human insulin was administered before each meal. LANTUS was administered at bedtime. NPH insulin was administered either as once daily at bedtime or in the morning and at bedtime when used twice daily.
In Study A, the average age was 39 years. The majority of patients were White (99%) and 56% were male. The mean BMI was approximately 24.9 kg/m2. The mean duration of diabetes was 16 years.
In Study B, the average age was 39 years. The majority of patients were White (95%) and 51% were male. The mean BMI was approximately 25.8 kg/m2. The mean duration of diabetes was 17 years.
In another clinical study (Study C), patients with type 1 diabetes (n=619) were randomized to 16 weeks of basal-bolus treatment with LANTUS or NPH insulin. Insulin lispro was used before each meal. LANTUS was administered once daily at bedtime and NPH insulin was administered once or twice daily. The average age was 39 years. The majority of patients were White (97%) and 51% were male. The mean BMI was approximately 25.6 kg/m2. The mean duration of diabetes was 19 years.
In these 3 adult studies, LANTUS and NPH insulin had similar effects on HbA1c (Table 9) with a similar overall rate of severe symptomatic hypoglycemia [see Adverse Reactions (6.1)].
Table 9: Type 1 Diabetes Mellitus – Adults Study A Study B Study C Treatment duration 28 weeks 28 weeks 16 weeks Treatment in combination with Regular insulin Regular insulin Insulin lispro LANTUS NPH LANTUS NPH LANTUS NPH Number of subjects treated 292 293 264 270 310 309 HbA1c Baseline HbA1c 8.0 8.0 7.7 7.7 7.6 7.7 Adjusted mean change at study end +0.2 +0.1 -0.2 -0.2 -0.1 -0.1 Treatment Difference (95% CI) +0.1 (0.0; +0.2) +0.1 (-0.1; +0.2) 0.0 (-0.1; +0.1) Basal insulin dose Baseline mean 21 23 29 29 28 28 Mean change from baseline -2 0 -4 +2 -5 +1 Total insulin dose Baseline mean 48 52 50 51 50 50 Mean change from baseline -1 0 0 +4 -3 0 Fasting blood glucose (mg/dL) Baseline mean 167 166 166 175 175 173 Adj. mean change from baseline -21 -16 -20 -17 -29 -12 Body weight (kg) Baseline mean 73.2 74.8 75.5 75.0 74.8 75.6 Mean change from baseline 0.1 -0.0 0.7 1.0 0.1 0.5 Pediatric Patients with Type 1 Diabetes
In a randomized, controlled clinical study (Study D), pediatric patients (age range 6 to 15 years) with type 1 diabetes (n=349) were treated for 28 weeks with a basal-bolus insulin regimen where regular human insulin was used before each meal. LANTUS was administered once daily at bedtime and NPH insulin was administered once or twice daily. The average age was 11.7 years. The majority of patients were White (97%) and 52% were male. The mean BMI was approximately 18.9 kg/m2. The mean duration of diabetes was 5 years. Similar effects on HbA1c (Table 10) were observed in both treatment groups [see Adverse Reactions (6.1)].
Table 10: Type 1 Diabetes Mellitus – Pediatric Patients Study D Treatment duration 28 weeks Treatment in combination with Regular insulin LANTUS + Regular insulin NPH + Regular insulin Number of subjects treated 174 175 HbA1c Baseline mean 8.5 8.8 Change from baseline (adjusted mean) +0.3 +0.3 Difference from NPH (adjusted mean) 0.0 (95% CI ) (-0.2; +0.3) Basal insulin dose Baseline mean 19 19 Mean change from baseline -1 +2 Total insulin dose Baseline mean 43 43 Mean change from baseline +2 +3 Fasting blood glucose (mg/dL) Baseline mean 194 191 Mean change from baseline -23 -12 Body weight (kg) Baseline mean 45.5 44.6 Mean change from baseline 2.2 2.5 14.3 Clinical Studies in Adults with Type 2 Diabetes
In a randomized, controlled clinical study (Study E) in 570 adults with type 2 diabetes, LANTUS was evaluated for 52 weeks in combination with oral antidiabetic medications (a sulfonylurea, metformin, acarbose, or combinations of these drugs). The average age was 60 years old. The majority of patients were White (93%) and 54% were male. The mean BMI was approximately 29.1 kg/m2. The mean duration of diabetes was 10 years. LANTUS administered once daily at bedtime was as effective as NPH insulin administered once daily at bedtime in reducing HbA1c and fasting glucose (Table 11). The rate of severe symptomatic hypoglycemia was similar in LANTUS and NPH insulin treated patients [see Adverse Reactions (6.1)].
In a randomized, controlled clinical study (Study F), in adult patients with type 2 diabetes not using oral antidiabetic medications (n=518), a basal-bolus regimen of LANTUS once daily at bedtime or NPH insulin administered once or twice daily was evaluated for 28 weeks. Regular human insulin was used before meals, as needed. The average age was 59 years. The majority of patients were White (81%) and 60% were male. The mean BMI was approximately 30.5 kg/m2. The mean duration of diabetes was 14 years. LANTUS had similar effectiveness as either once- or twice-daily NPH insulin in reducing HbA1c and fasting glucose (Table 11) with a similar incidence of hypoglycemia [see Adverse Reactions (6.1)].
In a randomized, controlled clinical study (Study G), adult patients with type 2 diabetes were randomized to 5 years of treatment with once-daily LANTUS or twice-daily NPH insulin. For patients not previously treated with insulin, the starting dosage of LANTUS or NPH insulin was 10 units daily. Patients who were already treated with NPH insulin either continued on the same total daily NPH insulin dose or started LANTUS at a dosage that was 80% of the total previous NPH insulin dosage. The primary endpoint for this study was a comparison of the progression of diabetic retinopathy by 3 or more steps on the Early Treatment Diabetic Retinopathy Study (ETDRS) scale. HbA1c change from baseline was a secondary endpoint. Similar glycemic control in the 2 treatment groups was desired in order to not confound the interpretation of the retinal data. Patients or study personnel used an algorithm to adjust the LANTUS and NPH insulin dosages to a target fasting plasma glucose ≤100 mg/dL. After the LANTUS or NPH insulin dosage was adjusted, other antidiabetic agents, including premeal insulin were to be adjusted or added. The average age was 55 years. The majority of patients were White (85%) and 54% were male. The mean BMI was approximately 34.3 kg/m2. The mean duration of diabetes was 11 years. The LANTUS group had a smaller mean reduction from baseline in HbA1c compared to the NPH insulin group, which may be explained by the lower daily basal insulin doses in the LANTUS group (Table 11). The incidences of severe symptomatic hypoglycemia were similar between groups [see Adverse Reactions (6.1)].
Table 11: Type 2 Diabetes Mellitus – Adults Study E Study F Study G Treatment duration 52 weeks 28 weeks 5 years Treatment in combination with Oral agents Regular insulin Regular insulin LANTUS NPH LANTUS NPH LANTUS NPH - *
- In Study G, the baseline dose of basal or total insulin was the first available on-treatment dose prescribed during the study (on visit month 1.5).
Number of subjects treated 289 281 259 259 513 504 HbA1c Baseline mean 9.0 8.9 8.6 8.5 8.4 8.3 Adjusted mean change from baseline -0.5 -0.4 -0.4 -0.6 -0.6 -0.8 LANTUS – NPH -0.1 +0.2 +0.2 95% CI for Treatment difference (-0.3; +0.1) (0.0; +0.4) (+0.1; +0.4) Basal insulin dose* Baseline mean 14 15 44.1 45.5 39 44 Mean change from baseline +12 +9 -1 +7 +23 +30 Total insulin dose* Baseline mean 14 15 64 67 48 53 Mean change from baseline +12 +9 +10 +13 +41 +40 Fasting blood glucose (mg/dL) Baseline mean 179 180 164 166 190 180 Adj. mean change from baseline -49 -46 -24 -22 -45 -44 Body weight (kg) Baseline mean 83.5 82.1 89.6 90.7 100 99 Adj. mean change from baseline 2.0 1.9 0.4 1.4 3.7 4.8 14.4 Additional Clinical Studies in Adults with Diabetes Type 1 and Type 2
Different Timing of LANTUS Administration in Diabetes Type 1 and Diabetes Type 2
The safety and efficacy of once daily LANTUS administered either at pre-breakfast, pre-dinner, or at bedtime were evaluated in a randomized, controlled clinical study in adult patients with type 1 diabetes (Study H, n=378). Patients were also treated with insulin lispro at mealtime. The average age was 41 years. All patients were White (100%) and 54% were male. The mean BMI was approximately 25.3 kg/m2. The mean duration of diabetes was 17 years.
LANTUS administered at pre-breakfast or at pre-dinner (both once daily) resulted in similar reductions in HbA1c compared to that with bedtime administration (see Table 12). In these patients, data are available from 8-point home glucose monitoring. The maximum mean blood glucose was observed just prior to LANTUS injection regardless of time of administration. In this study, 5% of patients in the LANTUS-breakfast group discontinued treatment because of lack of efficacy. No patients in the other two groups (pre-dinner, bedtime) discontinued for this reason.
The safety and efficacy of once daily LANTUS administered pre-breakfast or at bedtime were also evaluated in a randomized, active-controlled clinical study (Study I, n=697) in patients with type 2 diabetes not adequately controlled on oral antidiabetic therapy. All patients in this study also received glimepiride 3 mg daily. The average age was 61 years. The majority of patients were White (97%) and 54% were male. The mean BMI was approximately 28.7 kg/m2. The mean duration of diabetes was 10 years. LANTUS given before breakfast was at least as effective in lowering HbA1c as LANTUS given at bedtime or NPH insulin given at bedtime (see Table 12).
Table 12: Study of Different Times of Once Daily LANTUS Dosing in Type 1 (Study H) and Type 2 (Study I) Diabetes Mellitus Treatment duration
Treatment in combination withStudy H
24 weeks
Insulin lisproStudy I
24 weeks
GlimepirideLANTUS
Before BreakfastLANTUS
Before DinnerLANTUS
BedtimeLANTUS
Before BreakfastLANTUS
BedtimeNPH
BedtimeNumber of subjects treated* 112 124 128 234 226 227 HbA1c Baseline mean 7.6 7.5 7.6 9.1 9.1 9.1 Mean change from baseline -0.2 -0.1 0.0 -1.3 -1.0 -0.8 Basal insulin dose (Units) Baseline mean 22 23 21 19 20 19 Mean change from baseline 5 2 2 11 18 18 Total insulin dose (Units) – – – NA† NA† NA† Baseline mean 52 52 49 – – – Mean change from baseline 2 3 2 – – – Body weight (kg) Baseline mean 77.1 77.8 74.5 80.7 82 81 Mean change from baseline 0.7 0.1 0.4 3.9 3.7 2.9 Progression of Retinopathy Evaluation in Adults with Diabetes Type 1 and Diabetes Type 2
LANTUS was compared to NPH insulin in a 5-year randomized clinical study that evaluated the progression of retinopathy as assessed with fundus photography using a grading protocol derived from the Early Treatment Diabetic Retinopathy Scale (ETDRS). Patients had type 2 diabetes (mean age 55 years) with no (86%) or mild (14%) retinopathy at baseline. Mean baseline HbA1c was 8.4%. The primary outcome was progression by 3 or more steps on the ETDRS scale at study endpoint. Patients with prespecified postbaseline eye procedures (pan-retinal photocoagulation for proliferative or severe nonproliferative diabetic retinopathy, local photocoagulation for new vessels, and vitrectomy for diabetic retinopathy) were also considered as 3-step progressors regardless of actual change in ETDRS score from baseline. Retinopathy graders were blinded to treatment group assignment.
The results for the primary endpoint are shown in Table 13 for both the per-protocol and intent-to-treat populations, and indicate similarity of LANTUS to NPH in the progression of diabetic retinopathy as assessed by this outcome. In this study, the numbers of retinal adverse events reported for LANTUS and NPH insulin treatment groups were similar for adult patients with type 1 and type 2 diabetes.
The ORIGIN Study of Major Cardiovascular Outcomes in Patients with Established CV Disease or CV Risk Factors
The Outcome Reduction with Initial Glargine Intervention study (i.e., ORIGIN) was an open-label, randomized, 2-by-2, factorial design study. One intervention in ORIGIN compared the effect of LANTUS to standard care on major adverse cardiovascular (CV) outcomes in 12,537 adults ≥ 50 years of age with:
- Abnormal glucose levels (i.e., impaired fasting glucose [IFG] and/or impaired glucose tolerance [IGT]) or early type 2 diabetes mellitus and
- Established CV disease or CV risk factors at baseline.
The objective of the study was to demonstrate that LANTUS use could significantly lower the risk of major CV outcomes compared to standard care. There were two coprimary composite CV endpoints:
- The first coprimary endpoint was the time to first occurrence of a major adverse CV event defined as the composite of CV death, nonfatal myocardial infarction, and nonfatal stroke.
- The second coprimary endpoint was the time to the first occurrence of CV death or nonfatal myocardial infarction or nonfatal stroke or revascularization procedure or hospitalization for heart failure.
Patients were randomized to either LANTUS (N=6,264) titrated to a goal fasting plasma glucose of ≤95 mg/dL or to standard care (N=6,273). Anthropometric and disease characteristics were balanced at baseline. The mean age was 64 years and 8% of patients were 75 years of age or older. The majority of patients were male (65%). Fifty nine percent were White, 25% were Latin, 10% were Asian and 3% were Black or African American. The median baseline BMI was 29 kg/m2. Approximately 12% of patients had abnormal glucose levels (IGT and/or IFG) at baseline and 88% had type 2 diabetes. For patients with type 2 diabetes, 59% were treated with a single oral antidiabetic drug, 23% had known diabetes but were on no antidiabetic drug and 6% were newly diagnosed during the screening procedure. The mean HbA1c (SD) at baseline was 6.5% (1.0). Fifty-nine percent of the patients had had a prior CV event and 39% had documented coronary artery disease or other CV risk factors.
Vital status was available for 99.9% and 99.8% of patients randomized to LANTUS and standard care respectively at end of study. The median duration of follow-up was 6.2 years (range: 8 days to 7.9 years). The mean HbA1c (SD) at the end of the study was 6.5% (1.1) and 6.8% (1.2) in the LANTUS and standard care group respectively. The median dose of LANTUS at end of study was 0.45 U/kg. Eighty-one percent of patients randomized to LANTUS were using LANTUS at end of the study. The mean change in body weight from baseline to the last treatment visit was 2.2 kg greater in the LANTUS group than in the standard care group.
Overall, the incidence of major adverse CV outcomes was similar between groups (see Table 14). All-cause mortality was also similar between groups.
Table 14: Cardiovascular Outcomes in ORIGIN in Patients with Established CV Disease or CV Risk Factors – Time to First Event Analyses LANTUS
N=6,264Standard Care
N=6,273LANTUS vs Standard Care n
(Events per 100 PY)n
(Events per 100 PY)Hazard Ratio (95% CI) Coprimary endpoints CV death, nonfatal myocardial infarction, or nonfatal stroke 1041
(2.9)1013
(2.9)1.02 (0.94, 1.11) CV death, nonfatal myocardial infarction, nonfatal stroke, hospitalization for heart failure or revascularization procedure 1792
(5.5)1727
(5.3)1.04 (0.97, 1.11) Components of coprimary endpoints CV death 580 576 1.00 (0.89, 1.13) Myocardial Infarction (fatal or nonfatal) 336 326 1.03 (0.88, 1.19) Stroke (fatal or nonfatal) 331 319 1.03 (0.89, 1.21) Revascularizations 908 860 1.06 (0.96, 1.16) Hospitalization for heart failure 310 343 0.90 (0.77, 1.05) In the ORIGIN study, the overall incidence of cancer (all types combined) or death from cancer (Table 15) was similar between treatment groups.
Table 15: Cancer Outcomes in ORIGIN – Time to First Event Analyses LANTUS
N=6,264Standard Care
N=6.273LANTUS vs Standard Care n
(Events per 100 PY)n
(Events per 100 PY)Hazard Ratio (95% CI) Cancer endpoints Any cancer event (new or recurrent) 559
(1.56)561
(1.56)0.99 (0.88, 1.11) New cancer events 524
(1.46)535
(1.49)0.96 (0.85, 1.09) Death due to Cancer 189
(0.51)201
(0.54)0.94 (0.77, 1.15) -
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
LANTUS (insulin glargine) injection is supplied as a clear and colorless solution containing 100 units/mL (U-100) available as follows:
LANTUS NDC number Package size 10 mL Multiple-dose vial 0088-2220-33 1 vial per carton 3 mL SoloStar single-patient-use prefilled pen 0088-2219-05 5 pens per carton Additional Information about LANTUS Solostar:
- The LANTUS SoloStar prefilled pen dials in 1-unit increments.
- Needles are not included in the packs.
Use BD Ultra-Fine® needles‡ with the SoloStar prefilled pens (these BD manufactured needles are sold separately).
16.2 Storage
Dispense in the original sealed carton with the enclosed Instructions for Use.
Store unused LANTUS in a refrigerator between 36°F and 46°F (2°C and 8°C). Do not freeze. Discard LANTUS if it has been frozen. Protect LANTUS from direct heat and light.
Storage conditions are summarized in the following table.
Not in-use (unopened) Not in-use (unopened) In-use (opened) Refrigerated Room Temperature (36°F–46°F [2°C–8°C]) (up to 86°F [30°C]) (see temperature below) 10 mL multiple-dose vial Until expiration date 28 days 28 days
Refrigerated or room temperature3 mL single-patient-use SoloStar prefilled pen Until expiration date 28 days 28 days
Room temperature only
(Do not refrigerate) -
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use). There are separate Instructions for Use for the Vial and LANTUS SoloStar Pen.
Never Share a LANTUS SoloStar Prefilled Pen or Insulin Syringe Between Patients
Advise patients that they must never share a LANTUS SoloStar pen with another person, even if the needle is changed. Advise patients using LANTUS vials not to re-use or share needles or insulin syringes with another person. Sharing carries a risk for transmission of blood-borne pathogens [see Warnings and Precautions (5.1)].
Hyperglycemia or Hypoglycemia
Inform patients that hypoglycemia is the most common adverse reaction with insulin. Inform patients of the symptoms of hypoglycemia (e.g., impaired ability to concentrate and react). This may present a risk in situations where these abilities are especially important, such as driving or operating other machinery. Advise patients who have frequent hypoglycemia or reduced or absent warning signs of hypoglycemia to use caution when driving or operating machinery [see Warnings and Precautions (5.3)].
Advise patients that changes in insulin regimen can predispose to hyperglycemia or hypoglycemia and that changes in insulin regimen should be made under close medical supervision [see Warnings and Precautions (5.2)].
Hypoglycemia Due to Medications Errors
Instruct patients to always check the insulin label before each injection to reduce the risk of a medication error [see Warnings and Precautions (5.4)].
Hypersensitivity Reactions
Advise patients that hypersensitivity reactions have occurred with LANTUS. Inform patients about the symptoms of hypersensitivity reactions [see Warnings and Precautions (5.5)].
-
SPL UNCLASSIFIED SECTION
Manufactured by:
sanofi-aventis U.S. LLC
Bridgewater, NJ 08807
A SANOFI COMPANYU.S. License No. 1752
©2023 sanofi-aventis U.S. LLC
LANTUS, TOUJEO and SoloStar are registered trademarks of sanofi-aventis U.S. LLC.
‡Other brands listed are the trademarks of their respective owners and are not trademarks of sanofi-aventis U.S. LLC
-
PATIENT PACKAGE INSERT
PATIENT INFORMATION
LANTUS® (LAN-tus)
(insulin glargine)
injection, for subcutaneous use
VIAL:100 units/mL (U-100)Do not share your syringes with other people, even if the needle has been changed. You may give other people a serious infection, or get a serious infection from them.
What is LANTUS?
LANTUS is a long-acting man-made-insulin used to control high blood sugar in adults and children with diabetes mellitus. LANTUS is not for use to treat diabetic ketoacidosis.Who should not use LANTUS?
Do not use LANTUS if you:- are having an episode of low blood sugar (hypoglycemia).
- have an allergy to insulin glargine or any of the ingredients in LANTUS. See the end of this Patient Information leaflet for a complete list of ingredients in LANTUS.
What should I tell my healthcare provider before using LANTUS?
Before using LANTUS, tell your healthcare provider about all your medical conditions including if you:- have liver or kidney problems.
- take other medicines, especially ones called TZDs (thiazolidinediones).
- have heart failure or other heart problems. If you have heart failure, it may get worse while you take TZDs with LANTUS.
- are pregnant, planning to become pregnant, or are breastfeeding. It is not known if LANTUS may harm your unborn baby or breastfeeding baby.
Before you start using LANTUS, talk to your healthcare provider about low blood sugar and how to manage it.How should I use LANTUS? - Read the detailed Instructions for Use that come with your LANTUS insulin.
- Use LANTUS exactly as your healthcare provider tells you to. Your healthcare provider should tell you how much LANTUS to use and when to use it.
- Know the amount of LANTUS you use. Do not change the amount of LANTUS you use unless your healthcare provider tells you to.
- Check your insulin label each time you give your injection to make sure you are using the correct insulin.
- Do not re-use needles. Always use a new needle for each injection. Re-use of needles increases your risk of having blocked needles, which may cause you to get the wrong dose of LANTUS. Using a new needle for each injection lowers your risk of getting an infection.
- You may take LANTUS at any time during the day but you must take it at the same time every day.
- Only use LANTUS that is clear and colorless. If your LANTUS is cloudy or slightly colored, return it to your pharmacy for a replacement.
- LANTUS is injected under the skin (subcutaneously) of your upper legs (thighs), upper arms, or stomach area (abdomen).
- Do not use LANTUS in an insulin pump or inject LANTUS into your vein (intravenously).
-
Change (rotate) injection sites within the area you chose with each dose to reduce your risk of getting lipodystrophy (pits in skin or thickened skin) and localized cutaneous amyloidosis (skin with lumps) at the injection sites.
- Do not use the exact same spot for each injection.
- Do not inject where the skin has pits, is thickened, or has lumps.
- Do not inject where the skin is tender, bruised, scaly or hard, or into scars or damaged skin.
- Do not mix LANTUS with any other type of insulin or liquid medicine.
- Check your blood sugar levels. Ask your healthcare provider what your blood sugar should be and when you should check your blood sugar levels.
Your dose of LANTUS may need to change because of: - a change in level of physical activity or exercise, weight gain or loss, increased stress, illness, change in diet, or because of the medicines you take.
What should I avoid while using LANTUS?
While using LANTUS do not:- drive or operate heavy machinery, until you know how LANTUS affects you.
- drink alcohol or use over-the-counter medicines that contain alcohol.
What are the possible side effects of LANTUS and other insulins?
LANTUS may cause serious side effects that can lead to death, including:-
low blood sugar (hypoglycemia). Signs and symptoms that may indicate low blood sugar include:
- dizziness or light-headedness, sweating, confusion, headache, blurred vision, slurred speech, shakiness, fast heartbeat, anxiety, irritability or mood change, hunger.
-
severe allergic reaction (whole body reaction). Get medical help right away if you have any of these signs or symptoms of a severe allergic reaction:
- a rash over your whole body, trouble breathing, a fast heartbeat, or sweating.
- low potassium in your blood (hypokalemia).
-
heart failure. Taking certain diabetes pills called TZDs (thiazolidinediones) with LANTUS may cause heart failure in some people. This can happen even if you have never had heart failure or heart problems before. If you already have heart failure it may get worse while you take TZDs with LANTUS. Your healthcare provider should monitor you closely while you are taking TZDs with LANTUS. Tell your healthcare provider if you have any new or worse symptoms of heart failure including:
- shortness of breath, swelling of your ankles or feet, sudden weight gain.
- trouble breathing; shortness of breath; fast heartbeat; swelling of your face, tongue, or throat; sweating; extreme drowsiness; dizziness; confusion.
- low blood sugar (hypoglycemia); weight gain; allergic reactions, including reactions at your injection site; skin thickening or pits at the injection site (lipodystrophy).
General information about the safe and effective use of LANTUS.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use LANTUS for a condition for which it was not prescribed. Do not give LANTUS to other people, even if they have the same symptoms that you have. It may harm them.
This Patient Information leaflet summarizes the most important information about LANTUS. If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about LANTUS that is written for healthcare professionals. For more information, go to www.lantus.com or call 1-800-633-1610.What are the ingredients in LANTUS? - Active ingredient: insulin glargine
- 10 mL vial inactive ingredients: glycerol 85%, m-cresol, polysorbate 20, zinc, and Water for Injection, USP. Hydrochloric acid and sodium hydroxide may be added to adjust the pH.
Manufactured by: sanofi-aventis U.S. LLC, Bridgewater, NJ 08807, A SANOFI COMPANY.
U.S. License No. 1752This Patient Information has been approved by the U.S. Food and Drug Administration.
Revised: June 2023
-
PATIENT PACKAGE INSERT
PATIENT INFORMATION
LANTUS® SOLOSTAR (LAN-tus)
(insulin glargine)
injection, for subcutaneous use
100 units/mL (U-100)Do not share your LANTUS SoloStar® pen with other people, even if the needle has been changed. You may give other people a serious infection, or get a serious infection from them.
What is LANTUS?
LANTUS is a long-acting man-made insulin used to control high blood sugar in adults and children with diabetes mellitus. LANTUS is not for use to treat diabetic ketoacidosis.Who should not use LANTUS?
Do not use LANTUS if you:- are having an episode of low blood sugar (hypoglycemia).
- have an allergy to insulin glargine or any of the ingredients in LANTUS. See the end of this Patient Information leaflet for a complete list of ingredients in LANTUS.
What should I tell my healthcare provider before using LANTUS?
Before using LANTUS, tell your healthcare provider about all your medical conditions including if you:- have liver or kidney problems.
- take other medicines, especially ones called TZDs (thiazolidinediones).
- have heart failure or other heart problems. If you have heart failure, it may get worse while you take TZDs with LANTUS.
- are pregnant, planning to become pregnant, or are breastfeeding. It is not known if LANTUS may harm your unborn baby or breastfeeding baby.
Before you start using LANTUS, talk to your healthcare provider about low blood sugar and how to manage it.How should I use LANTUS SoloStar? - Read the detailed Instructions for Use that come with your LANTUS SoloStar single-patient-use prefilled pen.
- Use LANTUS exactly as your healthcare provider tells you to. Your healthcare provider should tell you how much LANTUS to use and when to use it.
- Know the amount of LANTUS you use. Do not change the amount of LANTUS you use unless your healthcare provider tells you to.
- Check your insulin label each time you give your injection to make sure you are using the correct insulin.
- The dose counter on your SoloStar pen shows your dose of LANTUS. Do not make any dose changes unless your healthcare provider tells you to.
- Do not use a syringe to remove LANTUS from your SoloStar disposable prefilled pen.
- Do not re-use needles. Always use a new needle for each injection. Re-use of needles increases your risk of having blocked needles, which may cause you to get the wrong dose of LANTUS. Using a new needle for each injection lowers your risk of getting an infection. If your needle is blocked, follow the instructions in Step 3 of the Instructions for Use.
- You may take LANTUS at any time during the day but you must take it at the same time every day.
- LANTUS is injected under the skin (subcutaneously) of your upper legs (thighs), upper arms, or stomach area (abdomen).
- Do not use LANTUS in an insulin pump or inject LANTUS into your vein (intravenously).
-
Change (rotate) your injection sites within area you chose with each dose to reduce your risk of getting lipodystrophy (pits in skin or thickened skin) and localized cutaneous amyloidosis (skin with lumps) at the injection sites.
- Do not use the exact same spot for each injection.
- Do not inject where the skin has pits, is thickened, or has lumps.
- Do not inject where skin is tender, bruised, scaly or hard, or into scars or damaged skin.
- Do not mix LANTUS with any other type of insulin or liquid medicine.
- Check your blood sugar levels. Ask your healthcare provider what your blood sugar should be and when you should check your blood sugar levels.
Your dose of LANTUS may need to change because of: - a change in level of physical activity or exercise, weight gain or loss, increased stress, illness, change in diet, or because of the medicines you take.
What should I avoid while using LANTUS?
While using LANTUS do not:- drive or operate heavy machinery, until you know how LANTUS affects you.
- drink alcohol or use over-the-counter medicines that contain alcohol.
What are the possible side effects of LANTUS and other insulins?
LANTUS may cause serious side effects that can lead to death, including:-
low blood sugar (hypoglycemia). Signs and symptoms that may indicate low blood sugar include:
- dizziness or light-headedness, sweating, confusion, headache, blurred vision, slurred speech, shakiness, fast heartbeat, anxiety, irritability or mood change, hunger.
-
severe allergic reaction (whole body reaction). Get medical help right away if you have any of these signs or symptoms of a severe allergic reaction:
- a rash over your whole body, trouble breathing, a fast heartbeat, or sweating.
- low potassium in your blood (hypokalemia).
-
heart failure. Taking certain diabetes pills called TZDs (thiazolidinediones) with LANTUS may cause heart failure in some people. This can happen even if you have never had heart failure or heart problems before. If you already have heart failure it may get worse while you take TZDs with LANTUS. Your healthcare provider should monitor you closely while you are taking TZDs with LANTUS. Tell your healthcare provider if you have any new or worse symptoms of heart failure including:
- shortness of breath, swelling of your ankles or feet, sudden weight gain.
- trouble breathing; shortness of breath; fast heartbeat; swelling of your face, tongue, or throat; sweating; extreme drowsiness; dizziness; confusion.
- low blood sugar (hypoglycemia); weight gain; allergic reactions, including reactions at your injection site; skin thickening or pits at the injection site (lipodystrophy).
General information about the safe and effective use of LANTUS.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use LANTUS for a condition for which it was not prescribed. Do not give LANTUS to other people, even if they have the same symptoms that you have. It may harm them.
This Patient Information leaflet summarizes the most important information about LANTUS. If you would like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about LANTUS that is written for healthcare professionals. For more information about LANTUS call 1-800-633-1610 or go to the website www.lantus.com.What are the ingredients in LANTUS? - Active ingredient: insulin glargine
- 3 mL SoloStar prefilled pen inactive ingredients: glycerol 85%, m-cresol, zinc, and Water for Injection, USP. Hydrochloric acid and sodium hydroxide may be added to adjust the pH.
Manufactured by: sanofi-aventis U.S. LLC, Bridgewater, NJ 08807, A SANOFI COMPANY. U.S. License No. 1752 This Patient Information has been approved by the U.S. Food and Drug Administration.
Revised: June 2023 -
INSTRUCTIONS FOR USE
INSTRUCTIONS FOR USE
LANTUS® (LAN-tus)
(insulin glargine)
injection, for subcutaneous use
VIAL: 100 units/mL (U-100)These Instructions for Use contain information on how to inject LANTUS using the vial. Read these Instructions for Use before you start taking LANTUS and each time you get a new LANTUS vial. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or your treatment.
Do not share your LANTUS syringes with other people, even if the needle has been changed. You may give other people a serious infection, or get a serious infection from them.
Supplies Needed to Give Your Injection:
- a LANTUS 10 mL vial
- a U-100 insulin syringe and needle
- 2 alcohol swabs
- 1 sharps container for throwing away used needles and syringes. See "Disposing of used needles and syringes" at the end of these instructions.
Preparing to Inject LANTUS:
- Wash your hands with soap and water or clean your hands with alcohol.
- Check the LANTUS label to make sure you are taking the right type of insulin. This is especially important if you use more than 1 type of insulin.
- Check the LANTUS in the vial to make sure it is clear and colorless. Do not use LANTUS if it is colored or cloudy, or if you see particles in the solution.
- Do not use LANTUS after the expiration date stamped on the label or 28 days after you first use it.
- Always use a syringe that is marked for U-100 insulin. If you use a syringe other than a U-100 insulin syringe, you may get the wrong dose of LANTUS.
- Always use a new syringe and a new needle for each injection to help prevent infections and prevent blocked needles.
Step 1:
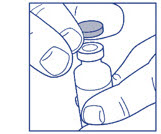
If you are using a new LANTUS vial, remove the protective cap. Do not remove the stopper.
Step 2:
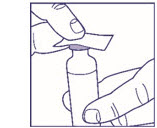
Wipe the top of the vial with an alcohol swab. You do not have to shake the vial of LANTUS before use.
Step 3:
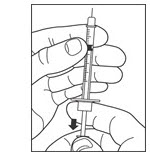
Draw air into the syringe equal to your LANTUS dose. Put the needle through the rubber top of the vial and push the plunger to inject the air into the vial.
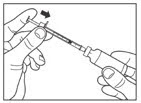
Step 4:
Leave the syringe in the vial and turn both upside down. Hold the syringe and vial firmly in one hand. Make sure the tip of the needle is in the LANTUS solution. With your free hand, pull the plunger to withdraw the correct dose into the syringe.
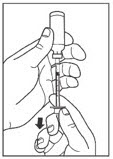
Step 5:
Before you take the needle out of the vial, check the syringe for air bubbles. If bubbles are in the syringe, hold the syringe straight up and tap the side of the syringe until the bubbles float to the top. Push the bubbles out with the plunger and draw insulin back in until you have the correct dose.
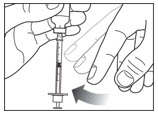
Step 6:
Remove the needle from the vial. Do not let the needle touch anything. You are now ready to inject.
Injecting LANTUS
- Inject your LANTUS (with a syringe) exactly as your healthcare provider has shown you.
- LANTUS is injected once daily at any time of the day but at the same time every day.
Step 7:
Choose your injection site:
- LANTUS is injected under the skin (subcutaneously) of your upper arms, thighs, or stomach area (abdomen).
- Change (rotate) your injection sites within the area you choose for each dose to reduce your risk of getting lipodystrophy (pits in the skin or thickened skin) and localized cutaneous amyloidosis (skin with lumps) at the injection sites.
- Do not inject where the skin has pits, is thickened, or has lumps.
- Do not inject where the skin is tender, bruised, scaly or hard, or into scars or damaged skin.
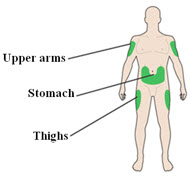
- Wipe the skin with an alcohol swab to clean the injection site. Let the injection site dry before you inject your dose.
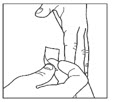
Step 8:
- Pinch the skin.
- Insert the needle under the skin in the way your healthcare provider showed you.
- Release the skin.
- Slowly push in the plunger of the syringe all the way, making sure you have injected all the LANTUS.
- Leave the needle in the skin for about 10 seconds.
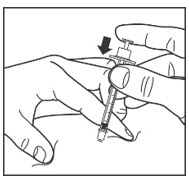
Step 9:
- Pull the needle straight out of your skin.
- Gently press the injection site for several seconds. Do not rub the area.
- Do not recap the used needle. Recapping the needle can lead to a needle-stick injury.
Disposing of Used Needles and Syringes
- Put your used needles and syringes in a FDA-cleared sharps disposal container right away after use. Do not throw away (dispose of) loose needles and syringes in your household trash.
- If you do not have a FDA-cleared sharps container, you may use a household container that is:
- made of a heavy-duty plastic,
- can be closed with a tight-fitting, puncture-resistant lid, without sharps being able to come out,
- upright and stable during use,
- leak resistant, and
- properly labeled to warn of hazardous waste inside the container.
- When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container. There may be state or local laws about how you should throw away used needles and syringes. For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA's website at: http://www.fda.gov/safesharpsdisposal.
- Do not dispose of your used sharps disposal container in your household trash unless your community guidelines permit this. Do not recycle your used sharps disposal container.
Storing and Disposing LANTUS?
Unopened (not in-use) LANTUS vials
- Store unused LANTUS vials in the refrigerator from 36°F to 46°F (2°C to 8°C).
- Do not freeze LANTUS.
- Keep LANTUS away from direct heat and light.
- If a vial has been frozen or overheated, throw it away.
- Unopened vials can be used until the expiration date on the carton and vial label if they have been stored in the refrigerator (they can be stored past 28 days in the refrigerator).
- Unopened vials should be thrown away after 28 days if they are stored at room temperature.
After LANTUS vials have been opened (in-use)
- Store in-use (opened) LANTUS vials in a refrigerator from 36°F to 46°F (2°C to 8°C) or at room temperature below 86°F (30°C) for up to 28 days.
- Do not freeze LANTUS. If a vial has been frozen, throw it away.
- Keep LANTUS out of direct heat and light.
- The LANTUS vial you are using should be thrown away after 28 days or if the expiration date has passed, even if it still has insulin left in it.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Manufactured by:
sanofi-aventis U.S. LLC
Bridgewater, NJ 08807
A SANOFI COMPANY
U.S. License No. 1752
©2022 sanofi-aventis U.S. LLC.Revised: June 2023
-
INSTRUCTIONS FOR USE
INSTRUCTIONS FOR USE
LANTUS® SOLOSTAR® (LAN-tus)
(insulin glargine)
injection, for subcutaneous use
3 mL Single-Patient-Use PREFILLED PEN: 100 units/mL (U-100)Read these Instructions for Use before you start taking the LANTUS Solostar pen and each time you get a new LANTUS SoloStar pen. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or your treatment.
Do not share your LANTUS SoloStar pen with other people, even if the needle has been changed. You may give other people a serious infection, or get a serious infection from them.
People who are blind or have vision problems should not use the LANTUS SoloStar prefilled pen without help from a person trained to use the LANTUS SoloStar prefilled pen.
LANTUS SoloStar is a disposable prefilled pen used to inject LANTUS. Each LANTUS SoloStar pen has 300 units of insulin which can be used for multiple injections. You can select doses from 1 to 80 units in steps of 1 unit. The pen plunger moves with each dose. The plunger will only move to the end of the cartridge when 300 units of LANTUS have been given.
Important Information You Need to Know Before Injecting LANTUS
- Do not use your pen if it is damaged or if you are not sure that it is working properly.
- Do not use a syringe to remove LANTUS from your pen.
- Do not reuse needles. If you do, you might get the wrong dose of LANTUS and/or increase the chance of getting an infection.
- Always perform a safety test (see Step 3).
- Always carry a spare pen and spare needles in case they got lost or stop working.
- Change (rotate) your injection sites within the area you choose for each dose (see "Places to inject").
Learn to Inject
- Talk with your healthcare provider about how to inject before using your pen.
- Ask for help if you have problems handling the pen, for example if you have problems with your sight.
- Read all these instructions before using your pen. If you do not follow all these instructions, you may get too much or too little insulin.
Need Help?
If you have any questions about your pen or about diabetes, ask your healthcare provider, or go to www.Lantus.com or call sanofi-aventis at 1-800-633-1610.
Extra Items You Will Need
- a new sterile needle (see Step 2).
- an alcohol swab.
- a puncture-resistant container for used needles and pens. (See "Throwing your pen away")
- Inject your insulin exactly as your healthcare provider has shown you.
- Inject your insulin under the skin (subcutaneously) of your upper legs (thighs), upper arms, or stomach area (abdomen).
- Change (rotate) your injection sites within the area you choose for each dose to reduce your risk of getting lipodystrophy (pits in skin or thickened skin) and localized cutaneous amyloidosis (skin with lumps) at the injection sites.
- Do not inject where the skin has pits, is thickened, or has lumps.
- Do not inject where the skin is tender, bruised, scaly or hard, or into scars or damaged skin.
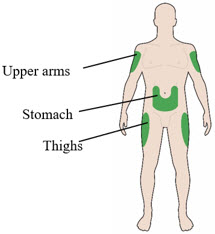
Get to know your pen
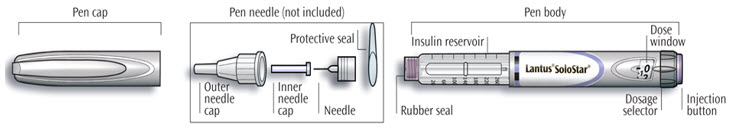
Step 1: Check your pen
Take a new pen out of the refrigerator at least 1 hour before you inject. Cold insulin is more painful to inject.
1A Check the name and expiration date on the label of your pen.
- Make sure you have the correct insulin.
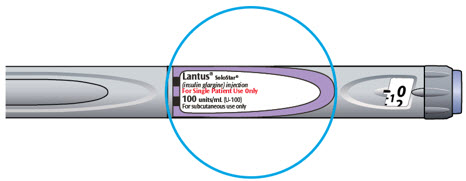
-
Do not use your pen after the expiration date.
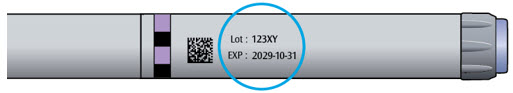
1B Pull off the pen cap.
1C Check that the insulin is clear.
-
Do not use the pen if the insulin looks cloudy, colored or contains particles.
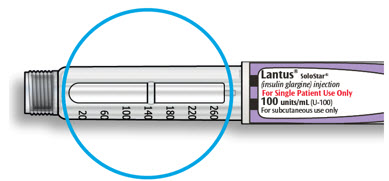
1D Wipe the rubber seal with an alcohol swab.
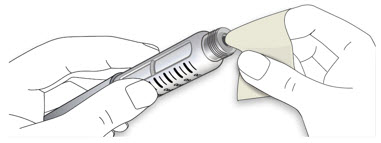
If you have other injector pens:
- Making sure you have the correct medicine is especially important if you have other injector pens.
- Do not reuse needles. Always use a new sterile needle for each injection. This helps stop blocked needles, contamination, and infection.
Only use needles* that are compatible for use with LANTUS SoloStar, such as BD Ultra-Fine®.2A Take a new needle and peel off the protective seal.
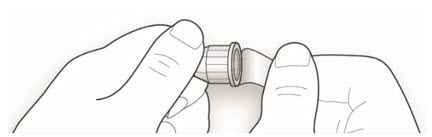
2B Keep the needle straight and screw it onto the pen until fixed. Do not over-tighten.
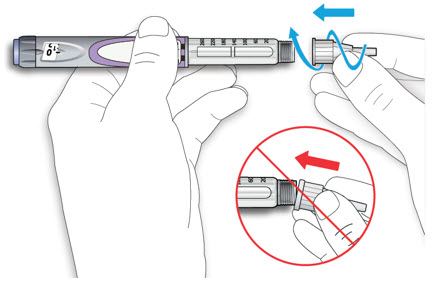
2C Pull off the outer needle cap. Keep this for later.
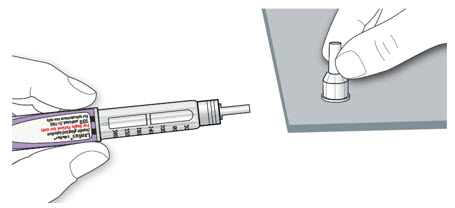
2D Pull off the inner needle cap and throw away.
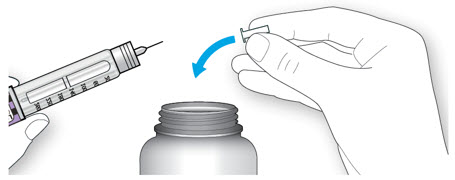
Handling needles
- Take care when handling needles to prevent needle-stick injury and cross-infection.
Always do a safety test before each injection to:
- Check your pen and the needle to make sure they are working properly.
- Make sure that you get the correct LANTUS dose.
3A Select 2 units by turning the dose selector until the dose pointer is at the 2 mark.
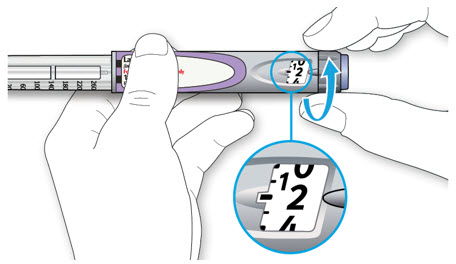
3B Press the injection button all the way in.
When insulin comes out of the needle tip, your pen is working correctly:
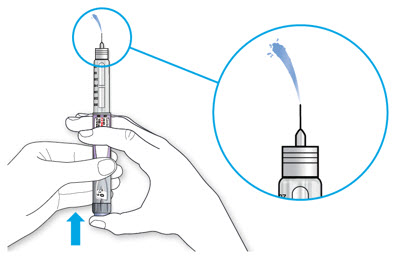
If no insulin appears:
- You may need to repeat this step up to 3 times before seeing insulin.
- If no insulin comes out after the third time, the needle may be blocked. If this happens:
- Do not use your pen if there is still no insulin coming out of the needle tip. Use a new pen.
- Do not use a syringe to remove insulin from your pen.
If you see air bubbles:
- You may see air bubbles in the insulin. This is normal, they will not harm you.
Do not select a dose or press the injection button without a needle attached. This may damage your pen.
4A Make sure a needle is attached and the dose is set to "0."
4B Turn the dose selector until the dose pointer lines up with your dose.
- If you turn past your dose, you can turn back down.
- If there are not enough units left in your pen for your dose, the dose selector will stop at the number of units left.
- If you cannot select your full prescribed dose, use a new pen or inject the remaining units and use a new pen to complete your dose.
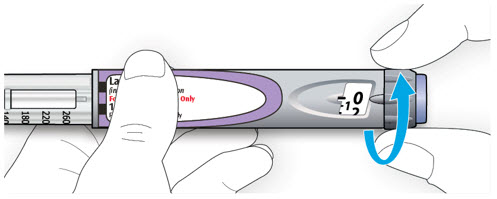
How to read the dose window
Even numbers are shown in line with dose pointer.
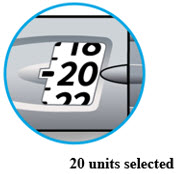
Odd numbers are shown as a line between even numbers.
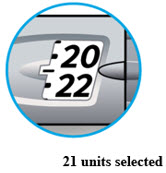
Units of LANTUS in your pen:
- Your pen contains a total of 300 units of LANTUS. You can select doses from 1 to 80 units in steps of 1 unit. Each pen contains more than 1 dose.
- You can see roughly how many units of insulin are left by looking at where the plunger is on the insulin scale.
Step 5: Injecting Your LANTUS Dose
If you find it hard to press the injection button in, do not force it as this may break your pen. See the section below for help.
5A Choose a place to inject as shown in the picture above.
5B Push the needle into your skin as shown by your healthcare provider.
Do not touch the injection button yet.
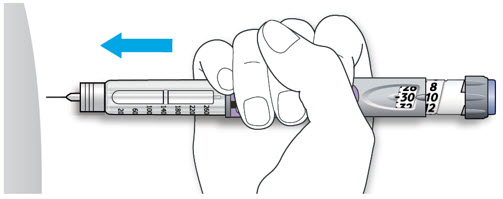
5C Place your thumb on the injection button. Then press all the way in and hold.
-
Do not press at an angle. Your thumb could block the dose selector from turning.
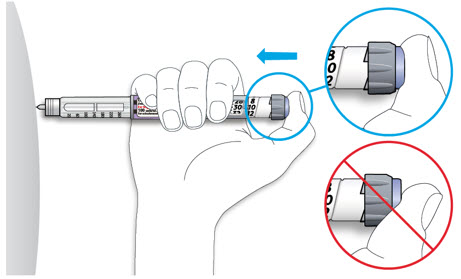
5D Keep the injection button held in and when you see "0" in the dose window, slowly count to 10.
- This will make sure you get your full dose.
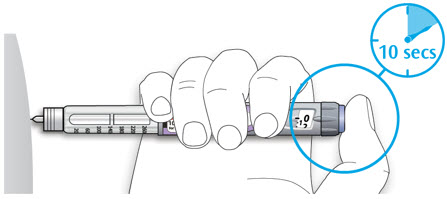
5E After holding and slowly counting to 10, release the injection button. Then remove the needle from your skin.
If you find it hard to press the button in:
- Change the needle (see Step 6 and Step 2) then do a safety test (see Step 3).
- If you still find it hard to press in, get a new pen.
- Do not use a syringe to remove insulin from your pen.
- Take care when handling needles to prevent needle-stick injury and cross-infection.
- Do not put the inner needle cap back on.
6A Grip the widest part of the outer needle cap. Keep the needle straight and guide it into the outer needle cap. Then push firmly on.
- The needle can puncture the cap if it is recapped at an angle.
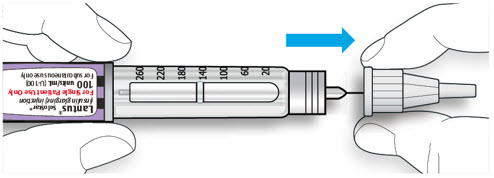
6B Grip and squeeze the widest part of the outer needle cap. Turn your pen several times with your other hand to remove the needle.
- Try again if the needle does not come off the first time.
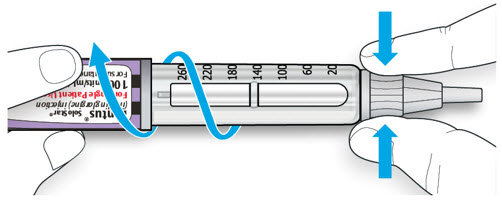
6C Throw away the used needle in a puncture-resistant container (see "Throwing your pen away" at the end of this Instructions for Use).
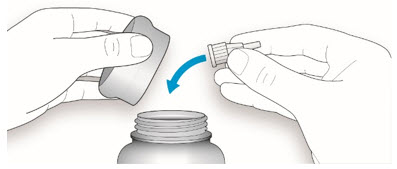
6D Put your pen cap back on.
- Do not put the pen back in the refrigerator.
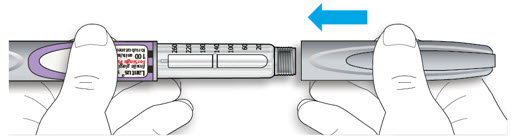
Storing the LANTUS Solostar Pen
Before first use
- Keep new pens in the refrigerator between 36°F to 46°F (2°C to 8°C).
- Do not freeze. Do not use LANTUS if it has been frozen.
After first use
- Keep your pen at room temperature below 86°F (30°C).
- Keep your pen away from heat or light.
- Store your pen with the pen cap on.
- Do not put your pen back in the refrigerator.
- Do not store your pen with the needle attached.
- Keep out of the reach of children.
- Only use your pen for up to 28 days after its first use. Throw away the LANTUS SoloStar pen you are using after 28 days, even if it still has insulin left in it.
Caring for Your LANTUS SoloStar Pen
Handle your pen with care
- Do not drop your pen or knock it against hard surfaces.
- If you think that your pen may be damaged, do not try to fix it. Use a new one.
Protect your pen from dust and dirt
- You can clean the outside of your pen by wiping it with a damp cloth (water only). Do not soak, wash or lubricate your pen. This may damage it.
Throwing your pen away
- The used LANTUS SoloStar pen may be thrown away in your household trash after you have removed the needle.
- Put the used needle in an FDA-cleared sharps disposal container right away after use. Do not throw away (dispose of) the used needles in your household trash.
- If you do not have a FDA-cleared sharps disposal container, you may use a household container that is:
- –
- made of a heavy-duty plastic,
- –
- can be closed with a tight-fitting, puncture-resistant lid, without sharps being able to come out,
- –
- upright and stable during use,
- –
- leak-resistant, and
- –
- properly labeled to warn of hazardous waste inside the container.
- When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container. There may be state or local laws about how you should throw away used needles and syringes. For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA's website at: http://www.fda.gov/safesharpsdisposal.
- Do not dispose of your used sharps disposal container in your household trash unless your community guidelines permit this. Do not recycle your used sharps disposal container.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Manufactured by:
sanofi-aventis U.S. LLC
Bridgewater, NJ 08807
A SANOFI COMPANY
U.S. License No. 1752
©2023 sanofi-aventis U.S. LLC.*Other brands listed are the registered trademarks of their respective owners and are not trademarks of sanofi-aventis U.S. LLC.
Revised: June 2023
- PRINCIPAL DISPLAY PANEL - 10 mL Vial Carton
-
PRINCIPAL DISPLAY PANEL - 3 mL Pen Carton
NDC 0088-2219-05
RX ONLY
Lantus® SoloStar®
insulin glargine injectionFor Single Patient Use Only
100 units/mL (U-100)
Five 3 mL Prefilled Pens – Dispense in this sealed cartonSolution for injection in a disposable insulin delivery device
Do not mix with other insulins
For subcutaneous injection only
Use only if solution is clear and colorless with no particles visible
Use within 28 days after initial use
*Needles not included (see back panel)SANOFI
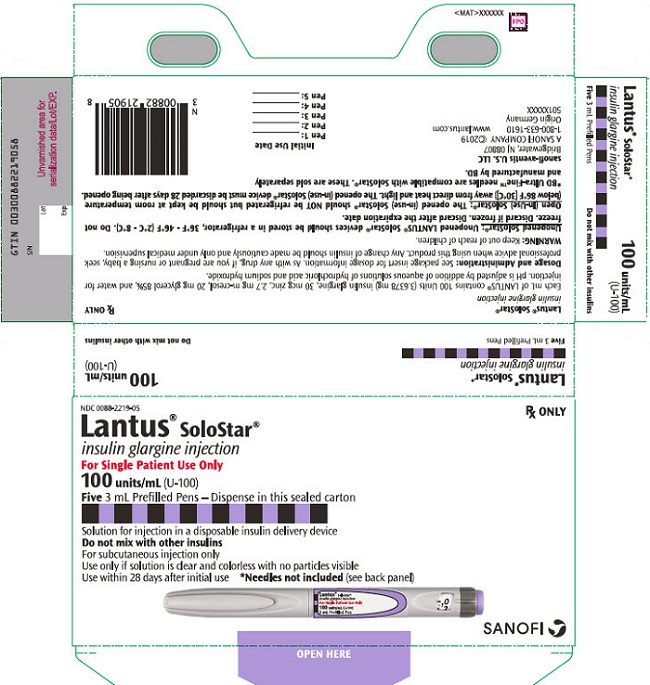
-
INGREDIENTS AND APPEARANCE
LANTUS
insulin glargine injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0088-2220 Route of Administration SUBCUTANEOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength INSULIN GLARGINE (UNII: 2ZM8CX04RZ) (INSULIN GLARGINE - UNII:2ZM8CX04RZ) INSULIN GLARGINE 100 [iU] in 1 mL Inactive Ingredients Ingredient Name Strength ZINC (UNII: J41CSQ7QDS) 30 ug in 1 mL METACRESOL (UNII: GGO4Y809LO) 2.7 mg in 1 mL GLYCERIN (UNII: PDC6A3C0OX) 20 mg in 1 mL WATER (UNII: 059QF0KO0R) HYDROCHLORIC ACID (UNII: QTT17582CB) SODIUM HYDROXIDE (UNII: 55X04QC32I) POLYSORBATE 20 (UNII: 7T1F30V5YH) 20 ug in 1 mL Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0088-2220-33 1 in 1 CARTON 09/23/2009 1 10 mL in 1 VIAL, GLASS; Type 0: Not a Combination Product 2 NDC:0088-2220-34 1 in 1 CARTON 09/23/2009 2 10 mL in 1 VIAL, GLASS; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA021081 09/23/2009 LANTUS SOLOSTAR
insulin glargine injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0088-2219 Route of Administration SUBCUTANEOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength INSULIN GLARGINE (UNII: 2ZM8CX04RZ) (INSULIN GLARGINE - UNII:2ZM8CX04RZ) INSULIN GLARGINE 100 [iU] in 1 mL Inactive Ingredients Ingredient Name Strength ZINC (UNII: J41CSQ7QDS) 30 ug in 1 mL METACRESOL (UNII: GGO4Y809LO) 2.7 mg in 1 mL GLYCERIN (UNII: PDC6A3C0OX) 20 mg in 1 mL WATER (UNII: 059QF0KO0R) HYDROCHLORIC ACID (UNII: QTT17582CB) SODIUM HYDROXIDE (UNII: 55X04QC32I) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0088-2219-00 1 in 1 CARTON 09/23/2009 1 3 mL in 1 SYRINGE; Type 3: Prefilled Biologic Delivery Device/System (syringe, patch, etc.) 2 NDC:0088-2219-05 5 in 1 CARTON 09/23/2009 2 NDC:0088-2219-01 3 mL in 1 SYRINGE; Type 3: Prefilled Biologic Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA021081 09/23/2009 Labeler - sanofi-aventis U.S. LLC (783243835) Establishment Name Address ID/FEI Business Operations Sanofi S.r.l. 338454274 ANALYSIS(0088-2220) , MANUFACTURE(0088-2220) , PACK(0088-2220) , LABEL(0088-2220) Establishment Name Address ID/FEI Business Operations Sanofi-Aventis Deutschland GmbH 313218430 ANALYSIS(0088-2219, 0088-2220) , MANUFACTURE(0088-2219, 0088-2220) , API MANUFACTURE(0088-2219, 0088-2220) , PACK(0088-2219, 0088-2220) , LABEL(0088-2219, 0088-2220)



