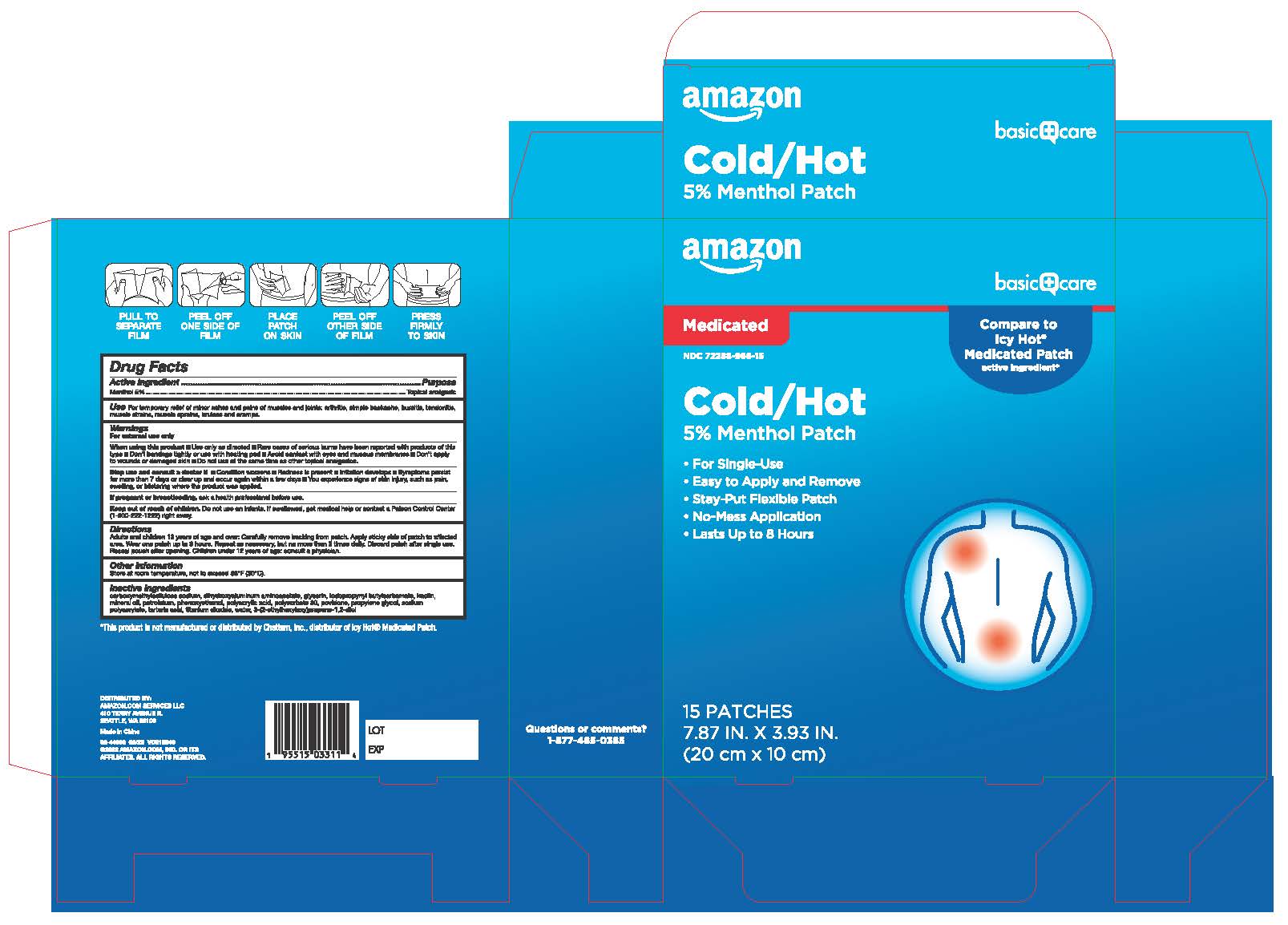
Label: AMAZON BASICS COLD AND HOT MEDICATED PATCH- menthol patch
- NDC Code(s): 72288-966-15
- Packager: Amazon.com Services LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated May 22, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- Purpose
- Use
-
Warnings
For external use only
When using this product
■Use only as directed ■ Rare cases of serious burns have been reported with products of this type ■Don't bandage tightly or use with heating pad ■ Avoid contact with eyes and mucous membranes ■Don't apply to wounds or damaged skin ■ Do not use at the same time as other topical analgesics.
-
Directions
Adults and children 12 years of age and over: Carefully remove backing from patch. Apply sticky side of patch to affected area. Wear one patch up to 8 hours. Repeat as necessary, but no more than 3 times daily. Discard patch after single use. Reseal pouch after opening. Children under 12 years of age: consult a physician.
- Other Information
-
Inactive Ingredients
carboxymethylcellulose sodium, dihydroxyaluminum aminoacetate, glycerin, iodopropynyl butylcarbamate, kaolin, mineral oil, petrolatum, phenoxyethanol, polyacrylic acid, polysorbate 80, povidone, propylene glycol, sodium polyacrylate, tartaric acid, titanium dioxide, water, 3-(2-ethylhexyloxy)propane-1,2-diol
- Amazon Basics Cold & Hot Medicated Patch
-
INGREDIENTS AND APPEARANCE
AMAZON BASICS COLD AND HOT MEDICATED PATCH
menthol patchProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:72288-966 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 5 g in 100 g Inactive Ingredients Ingredient Name Strength PHENOXYETHANOL (UNII: HIE492ZZ3T) WATER (UNII: 059QF0KO0R) POLYACRYLIC ACID (8000 MW) (UNII: 73861X4K5F) PETROLATUM (UNII: 4T6H12BN9U) CARBOXYMETHYLCELLULOSE SODIUM, UNSPECIFIED FORM (UNII: K679OBS311) POVIDONE (UNII: FZ989GH94E) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) IODOPROPYNYL BUTYLCARBAMATE (UNII: 603P14DHEB) KAOLIN (UNII: 24H4NWX5CO) POLYSORBATE 80 (UNII: 6OZP39ZG8H) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) DIHYDROXYALUMINUM AMINOACETATE (UNII: DO250MG0W6) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) GLYCERIN (UNII: PDC6A3C0OX) SODIUM POLYACRYLATE (8000 MW) (UNII: 285CYO341L) TARTARIC ACID (UNII: W4888I119H) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72288-966-15 15 in 1 CARTON 01/01/2023 1 1 g in 1 PATCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M017 01/01/2023 Labeler - Amazon.com Services LLC (128990418) Establishment Name Address ID/FEI Business Operations Foshan Aqua Gel Biotech Co., Ltd. 529128763 manufacture(72288-966)